Details of Drug-Drug Interaction
| Drug General Information (ID: DDIQH6L2VI) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Echothiophate (ophthalmic) | Drug Info | Neostigmine | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Ophthalmic Glaucoma Agents | Parasympathomimetics | |||||||
| Structure | |||||||||
| Mechanism of Echothiophate (ophthalmic)-Neostigmine Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Additive cholinergic effects Click to Show/Hide Mechanism Graph | |||||||||
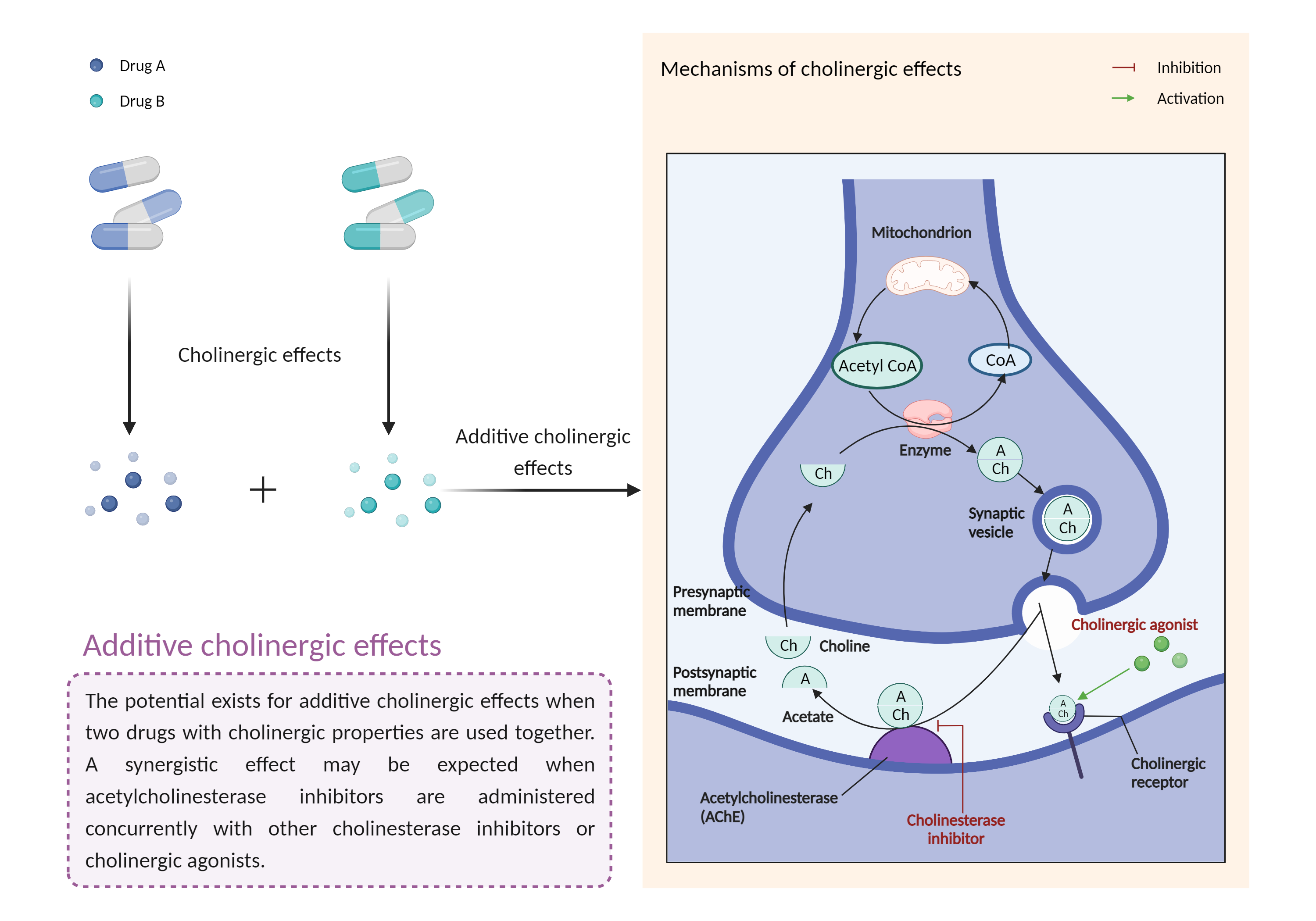 |
|||||||||
| Drug Name | Echothiophate (ophthalmic) | Neostigmine | |||||||
| Mechanism |
Cholinergic effects Acetylcholinesterase Inhibitor |
Cholinergic effects Acetylcholinesterase Inhibitor |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Acetylcholinesterase |
×
Structure
Sequence
MRPPQCLLHTPSLASPLLLLLLWLLGGGVGAEGREDAELLVTVRGGRLRGIRLKTPGGPVSAFLGIPFAEPPMGPRRFLPPEPKQPWSGVVDATTFQSVCYQYVDTLYPGFEGTEMWNPNRELSEDCLYLNVWTPYPRPTSPTPVLVWIYGGGFYSGASSLDVYDGRFLVQAERTVLVSMNYRVGAFGFLALPGSREAPGNVGLLDQRLALQWVQENVAAFGGDPTSVTLFGESAGAASVGMHLLSPPSRGLFHRAVLQSGAPNGPWATVGMGEARRRATQLAHLVGCPPGGTGGNDTELVACLRTRPAQVLVNHEWHVLPQESVFRFSFVPVVDGDFLSDTPEALINAGDFHGLQVLVGVVKDEGSYFLVYGAPGFSKDNESLISRAEFLAGVRVGVPQVSDLAAEAVVLHYTDWLHPEDPARLREALSDVVGDHNVVCPVAQLAGRLAAQGARVYAYVFEHRASTLSWPLWMGVPHGYEIEFIFGIPLDPSRNYTAEEKIFAQRLMRYWANFARTGDPNEPRDPKAPQWPPYTAGAQQYVSLDLRPLEVRRGLRAQACAFWNRFLPKLLSATDTLDEAERQWKAEFHRWSSYMVHWKNQFDHYSKQDRCSDL
|
|||||||
| Gene Name | ACHE | ||||||||
| Uniprot ID | ACES_HUMAN | ||||||||
| KEGG Pathway | hsa:43 | ||||||||
| Protein Family | Type-B carboxylesterase/lipase family | ||||||||
| Protein Function |
Hydrolyzes rapidly the acetylcholine neurotransmitter released into the synaptic cleft allowing to terminate the signal transduction at the neuromuscular junction. Role in neuronal apoptosis.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Patients should be monitored for excessive cholinergic effects and worsening of myasthenic symptoms. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Phospholine Iodide (echothiophate iodide ophthalmic). Wyeth Laboratories, Philadelphia, PA. | ||||||||||||||||||
