Details of Drug-Drug Interaction
| Drug General Information (ID: DDIQ50TMB9) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Gabapentin | Drug Info | Brexanolone | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Analgesics | Antidepressants | |||||||
| Structure | |||||||||
| Mechanism of Gabapentin-Brexanolone Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
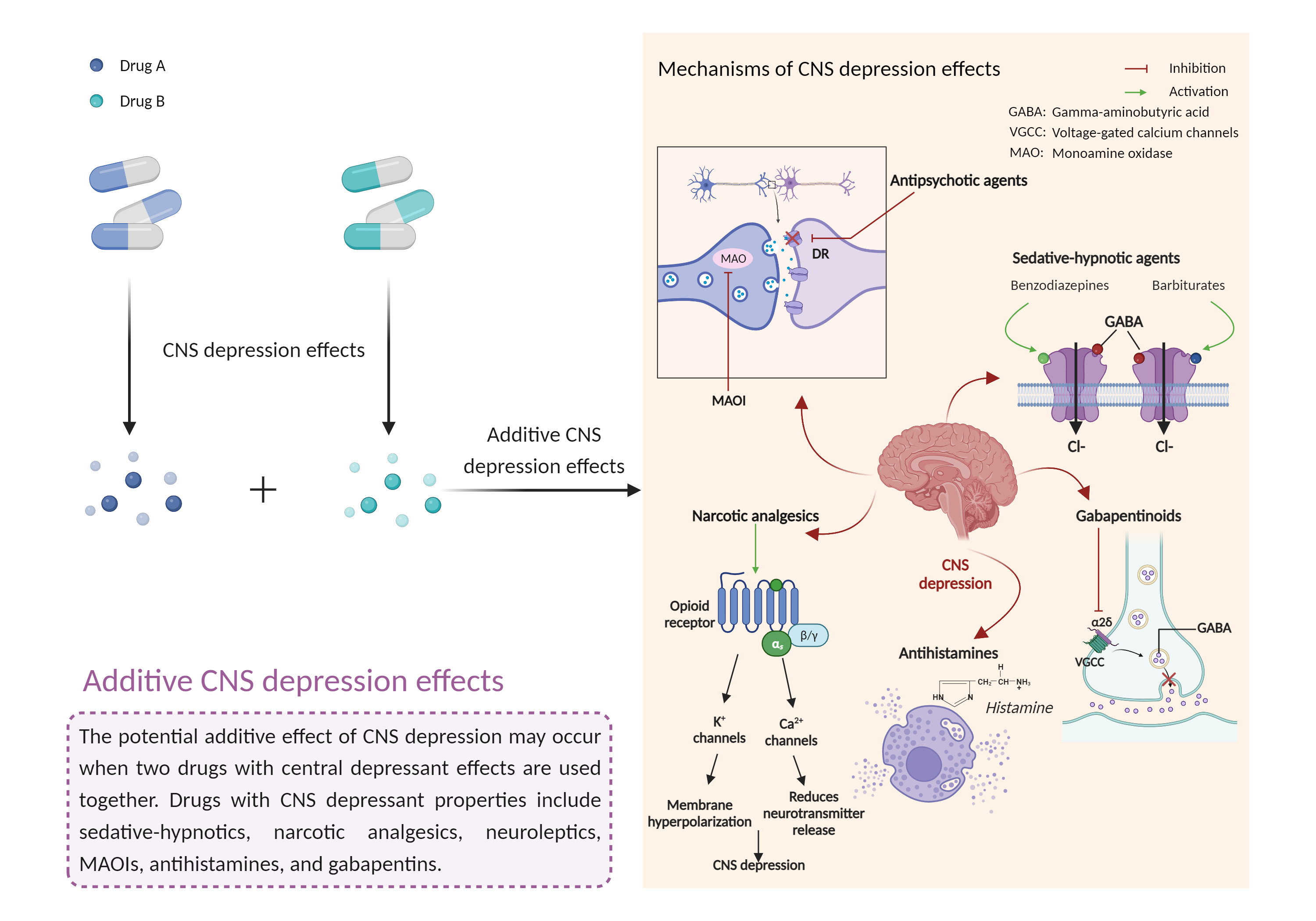
| Additive CNS depression effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Gabapentin | Brexanolone | |||||||
| Mechanism | CNS depression effects | CNS depression effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | CNS depression effects | ||||||||
| Factor Description | CNS depressants are drugs that inhibit or suppress brain activity and can reduce mental and physical processes. Excessive CNS depression can lead to decreased heart rate, slow breathing (less than 10 breaths per minute), extreme confusion or loss of memory, nausea and vomiting, poor judgment, blue lips or fingertips, irritability and aggression, and clammy or cold skin. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised during concomitant use of brexanolone with CNS depressants, antidepressants, or other agents that cause sedation. Patients should be closely monitored for excessive sedation and sudden loss of consciousness and have continuous pulse oximetry monitoring. During the brexanolone infusion, monitor for sedative effects every 2 hours during planned, non-sleep periods, and immediately stop the infusion if there are signs or symptoms of excessive sedation. After symptoms resolve, the infusion may be resumed at the same or lower dose as clinically appropriate. If pulse oximetry reveals hypoxia, immediately stop the infusion and do not resume infusion following resolution of the hypoxia. Patients should be cautioned against driving, operating machinery, or engaging in potentially hazardous activities requiring mental alertness and motor coordination until sedative effects of brexanolone and other concomitant medications have dissipated. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Zulresso (brexanolone). Sage Therapeutics, Inc., Cambridge, MA. | ||||||||||||||||||
