Details of Drug-Drug Interaction
| Drug General Information (ID: DDIQ3KCS6G) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Hepatitis A Vaccine | Drug Info | Trastuzumab deruxtecan | Drug Info | |||||
| Drug Type | Vaccine | Monoclonal antibody | |||||||
| Therapeutic Class | Vaccines | Antineoplastics/Her2 Inhibitors | |||||||
| Mechanism of Hepatitis A Vaccine-Trastuzumab deruxtecan Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
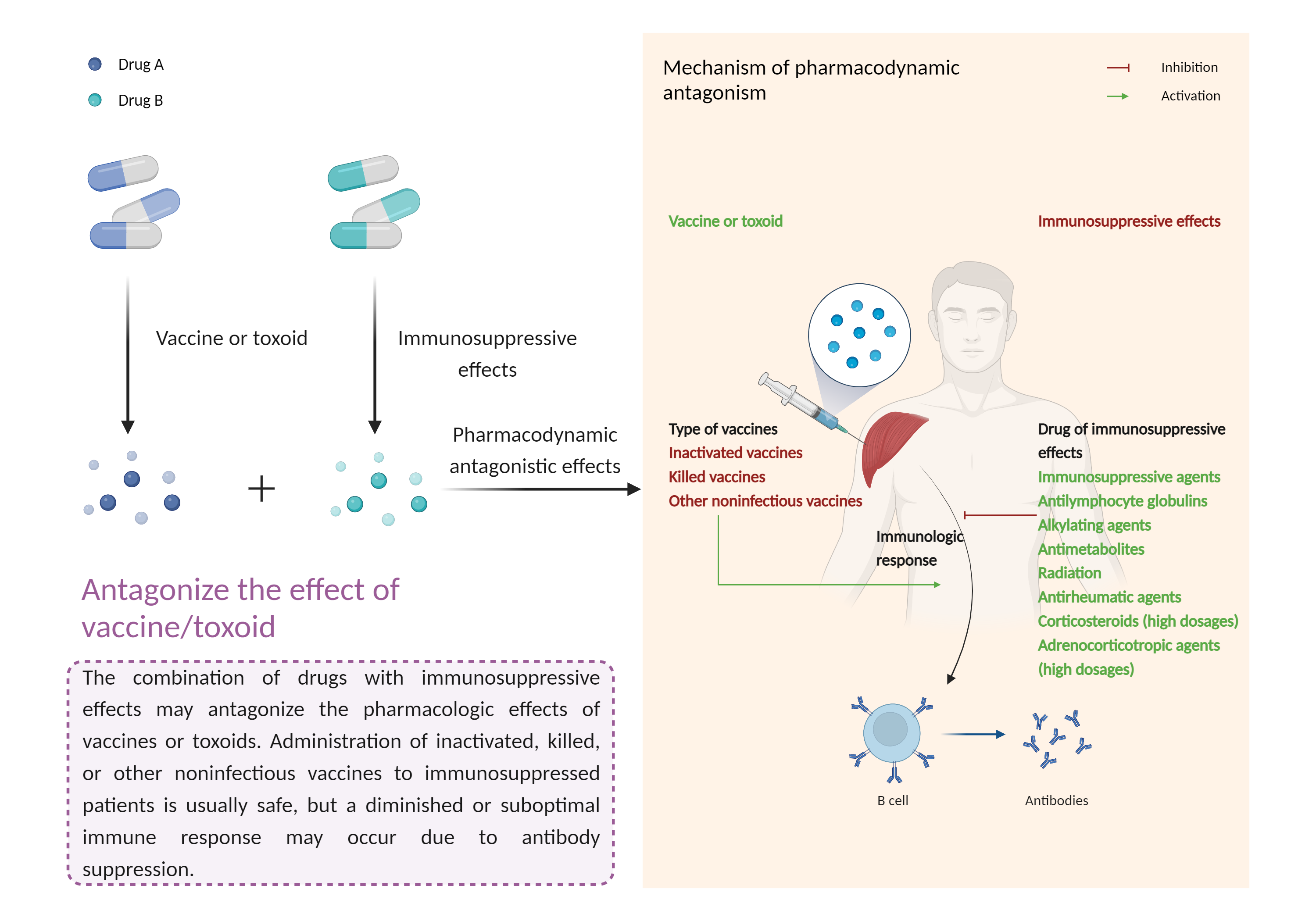
| Antagonize the effect of vaccine/toxoid Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Hepatitis A Vaccine | Trastuzumab deruxtecan | |||||||
| Mechanism | Vaccine or toxoid | Immunosuppressive effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Vaccine/toxoid | ||||||||
| Factor Description | The beneficial effects of the vaccine/toxinoid may be reduced, leading to a diminished or suboptimal immune response. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | In general, the US Department of Public Health Advisory Committee on Immunization Practices (ACIP) recommends that inactivated or killed vaccines be administered to non-HIV immunosuppressed patients according to the same guidelines as for healthy patients. For Haemophilus influenzae b vaccine, some experts recommend that it be administered at least 2 weeks before starting or 3 months after discontinuing chemotherapy when used in patients with Hodgkin's disease. For rabies vaccine, some authorities suggest that immunosuppressive agents should generally be avoided during postexposure therapy except when absolutely necessary for the treatment of other conditions. | ||||||||
