Details of Drug-Drug Interaction
| Drug General Information (ID: DDIPGU8LS2) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Dimethyl fumarate | Drug Info | Monomethyl fumarate | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Selective Immunosuppressants | Selective Immunosuppressants | |||||||
| Structure | |||||||||
| Mechanism of Dimethyl fumarate-Monomethyl fumarate Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
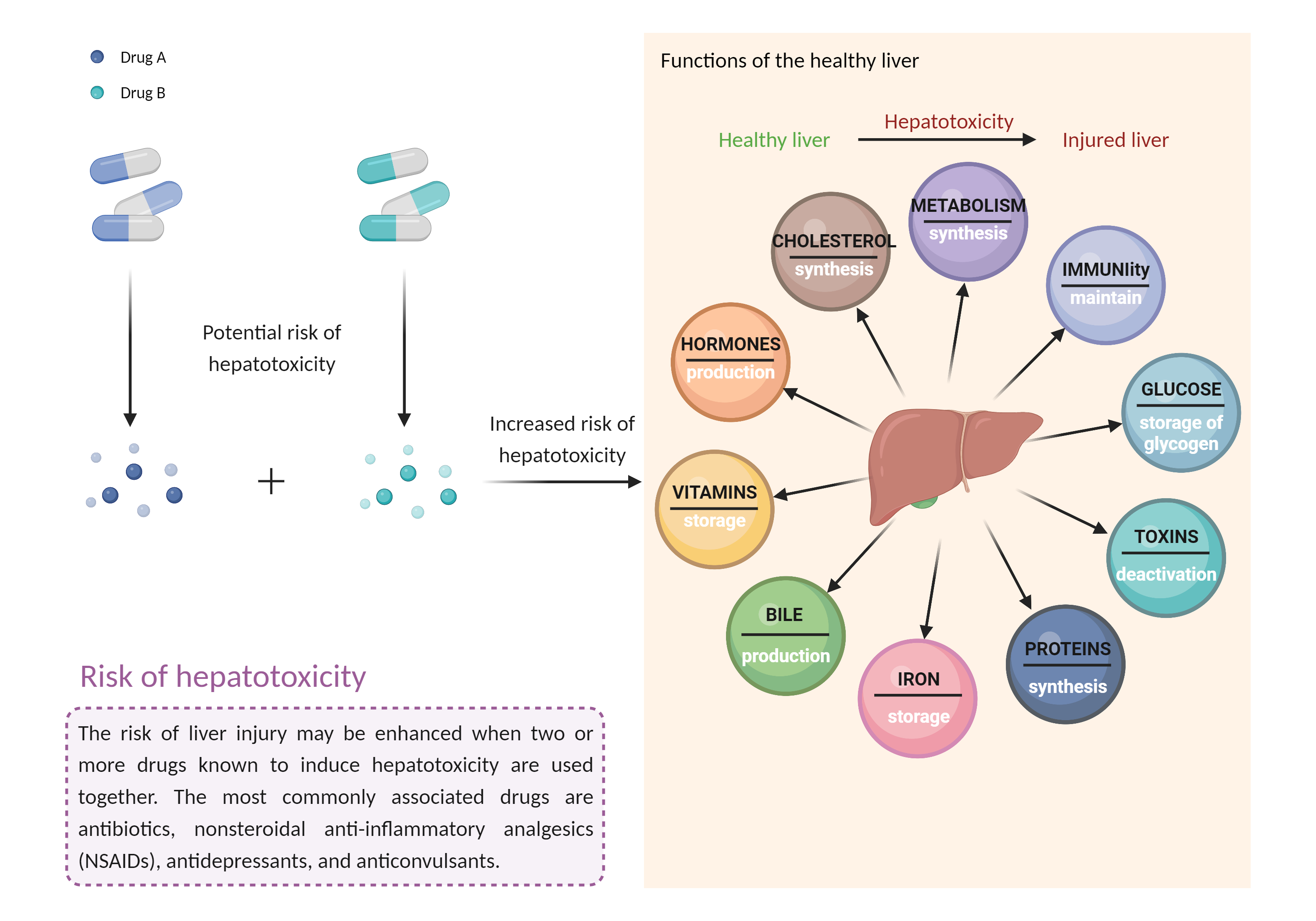
| Increased risk of hepatotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Dimethyl fumarate | Monomethyl fumarate | |||||||
| Mechanism | Hepatotoxicity | Hepatotoxicity | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hepatotoxicity | ||||||||
| Factor Description | Combination of drugs that can induce hepatotoxicity may increase the risk of liver injury. Symptoms vary depending on the level of exposure and the total extent of liver damage, and may cause few symptoms if the damage is mild, and eventually lead to liver failure in patients with severe damage. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Coadministration of monomethyl fumarate, dimethyl fumarate, or diroximel fumarate with one another is considered contraindicated. According to the prescribing information, treatment with monomethyl fumarate may be initiated the day following discontinuation of dimethyl fumarate or diroximel fumarate. Likewise, diroximel fumarate may be started the day after stopping dimethyl fumarate. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Tecfidera (dimethyl fumarate). Biogen Inc, Cambridge, MA. | ||||||||||||||||||
| 2 | Product Information. Vumerity (diroximel fumarate). Alkermes, Inc, Cambridge, MA. | ||||||||||||||||||
