Details of Drug-Drug Interaction
| Drug General Information (ID: DDIP3BRSQ0) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Insulin human (regular) | Drug Info | Stanozolol | Drug Info | |||||
| Drug Type | Hormones | Small molecule | |||||||
| Therapeutic Class | Insulin/Antidiabetic Agents | Androgens/Anabolic Steroids | |||||||
| Structure | |||||||||
| Mechanism of Insulin human (regular)-Stanozolol Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Increased risk of hypoglycemia Click to Show/Hide Mechanism Graph | |||||||||
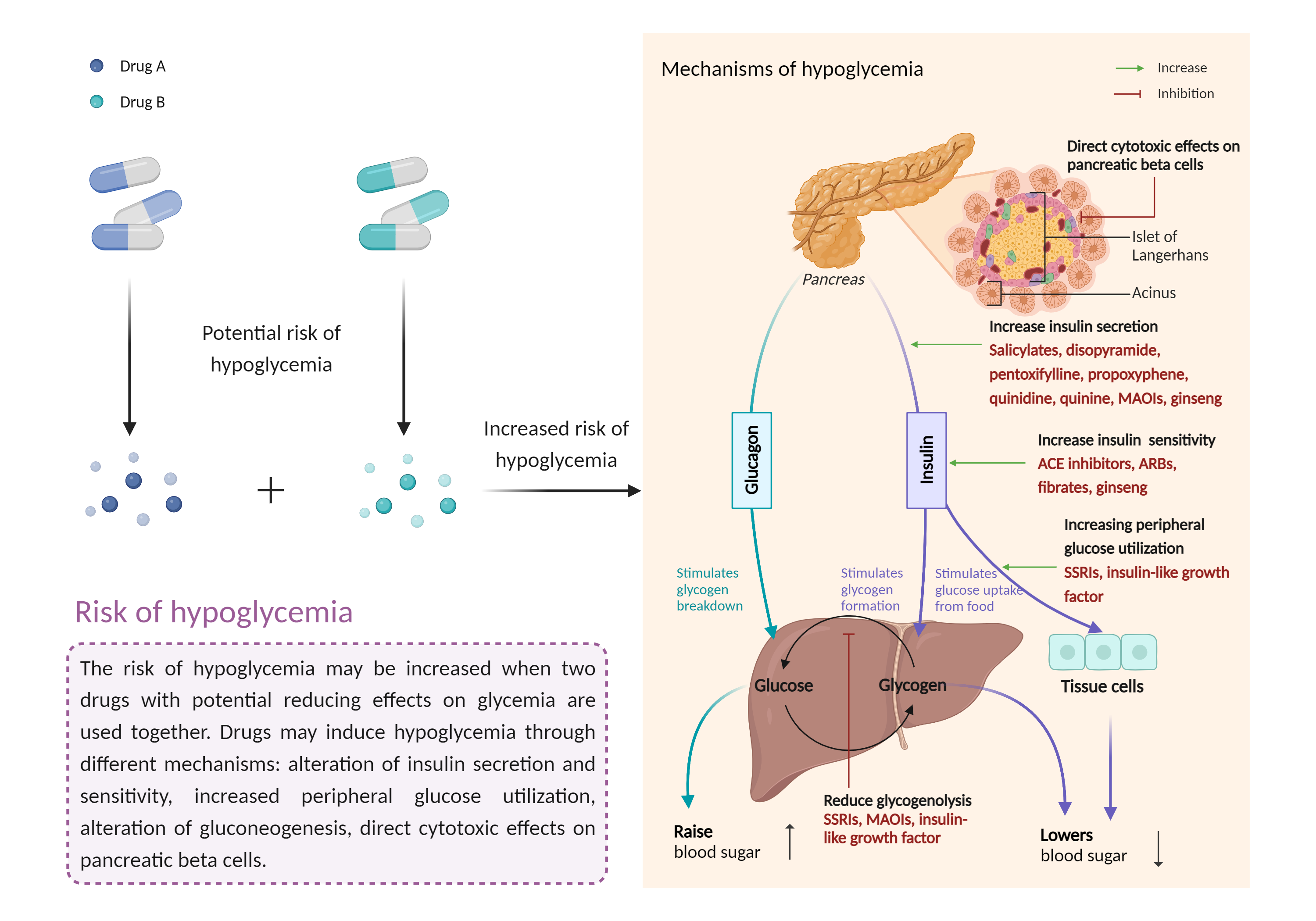 |
|||||||||
| Drug Name | Insulin human (regular) | Stanozolol | |||||||
| Mechanism | Hypoglycemic effects | Hypoglycemic effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hypoglycemia | ||||||||
| Factor Description | Hypoglycemia is a condition in which your blood sugar (glucose) level is below the standard range. It causes irregular or rapid heartbeat, pale skin, numbness of the lips, tongue or cheeks, and sweating. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Close monitoring for the development of hypoglycemia is recommended if these drugs are coadministered with insulin, particularly in patients with advanced age and/or renal impairment. The insulin dosage may require adjustment if an interaction is suspected. Patients should be apprised of the signs and symptoms of hypoglycemia (e.g., headache, dizziness, drowsiness, nausea, hunger, tremor, weakness, sweating, palpitations). | ||||||||
