Details of Drug-Drug Interaction
| Drug General Information (ID: DDIOR6NWYQ) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Vincristine | Drug Info | Guselkumab | Drug Info | |||||
| Drug Type | Small molecule | Monoclonal antibody | |||||||
| Therapeutic Class | Antineoplastics | Interleukin Inhibitors | |||||||
| Mechanism of Vincristine-Guselkumab Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
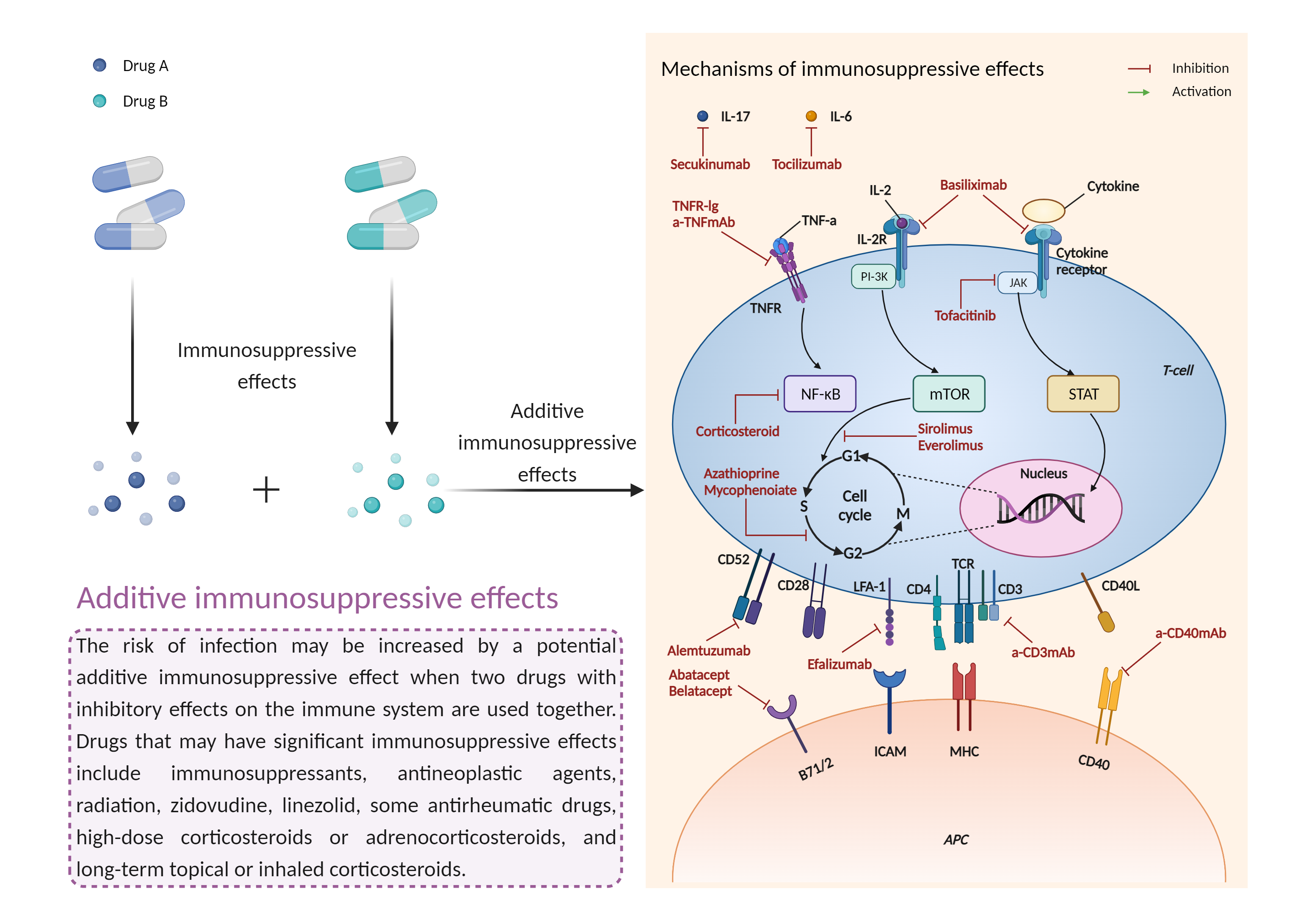
| Additive immunosuppressive effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Vincristine | Guselkumab | |||||||
| Mechanism 1 | Immunosuppressive effects | Immunosuppressive effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Immunosuppressive effects | ||||||||
| Factor Description | Immunosuppression is when your immune system is not functioning as it should. The immune system is made up of cells, tissues and organs that help the body fight off infections. If the immune system is suppressed, an infection that your body was able to control may become serious or even fatal. | ||||||||
| Mechanism Description |
|
||||||||
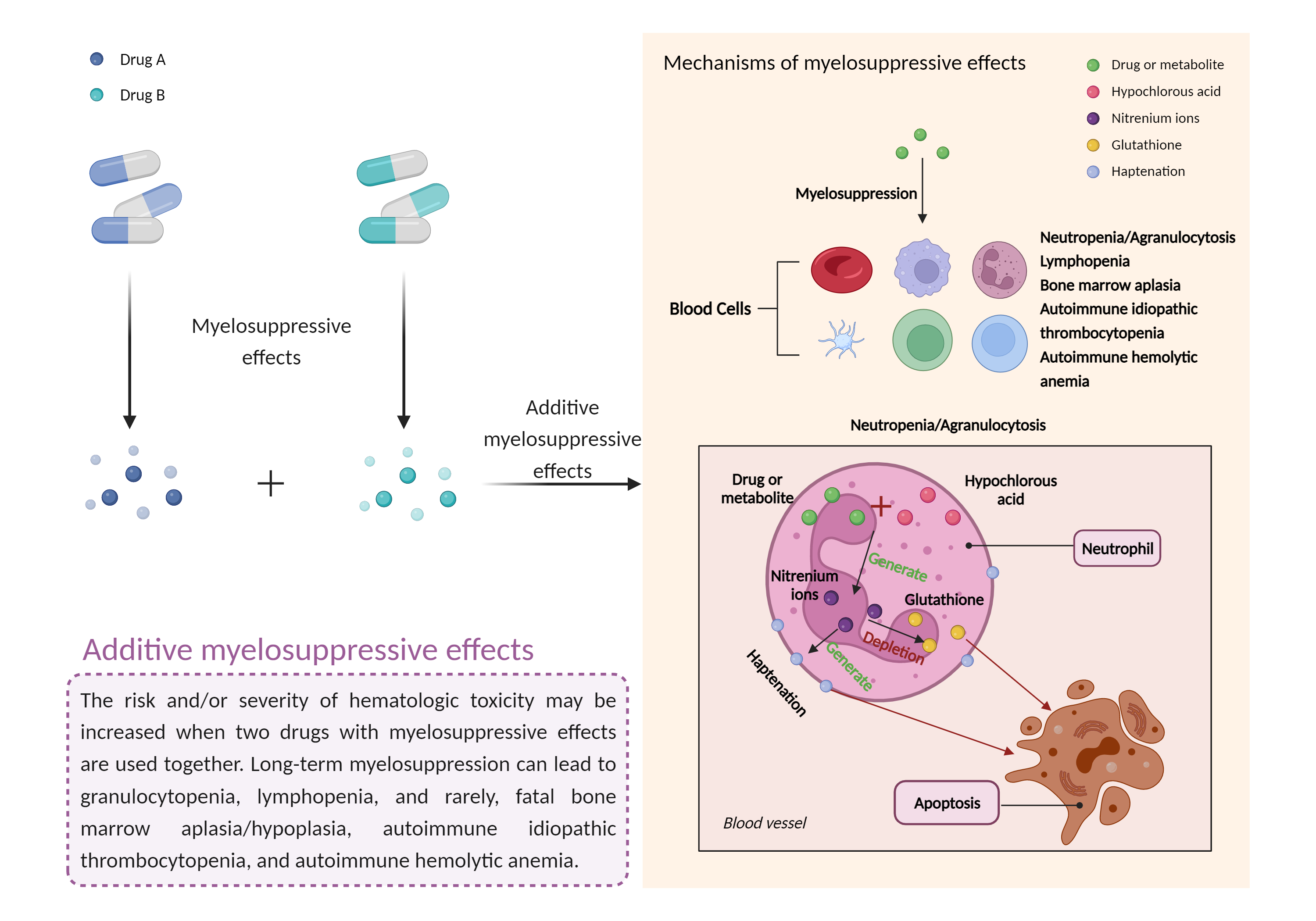
| Additive myelosuppressive effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Vincristine | Guselkumab | |||||||
| Mechanism 2 | Myelosuppressive effects | Myelosuppressive effects | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Myelosuppressive effects | ||||||||
| Factor Description | Myelosuppression, also known as bone marrow suppression, is a decrease in bone marrow activity that leads to a decrease in the production of blood cells. Some blood cell disorders include: erythrocytopenia (anemia), leukopenia (neutropenia), and thrombocytopenia (thrombocytopenia). | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Concomitant use of interleukin blockers with other immuno- or myelosuppressive agents should be avoided if possible. Caution is advised when interleukin inhibitors are prescribed to patients receiving concomitant drugs that are CYP450 substrates, particularly those with narrow therapeutic ranges such as immunosuppressants or antineoplastic agents. Clinical and/or laboratory monitoring should be considered following the initiation or withdrawal of interleukin inhibitor therapy, and the dosage(s) of these drugs adjusted accordingly. Clinicians should note that the effects of interleukin inhibitors on CYP450 activities may persist for several weeks after stopping therapy. | ||||||||
