Details of Drug-Drug Interaction
| Drug General Information (ID: DDIN94PR16) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Ibutilide | Drug Info | Orciprenaline | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiarrhythmic Agents | Adrenergic Bronchodilators | |||||||
| Structure | |||||||||
| Mechanism of Ibutilide-Orciprenaline Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Increased risk of prolong QT interval Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Ibutilide | Orciprenaline | |||||||
| Mechanism 1 | Prolong QT interval | Prolong QT interval | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | QT interval | ||||||||
| Factor Description | Long QT syndrome is a heart signaling disorder that can cause a fast, chaotic heartbeat (arrhythmia). Many people may not exhibit symptoms, and usually the condition is detected during routine medical tests. In others, the most common symptoms include: sudden fainting, palpitations, dizziness, seizures, sudden death. | ||||||||
| Mechanism Description |
|
||||||||
| Increased risk of ventricular arrhythmias Click to Show/Hide Mechanism Graph | |||||||||
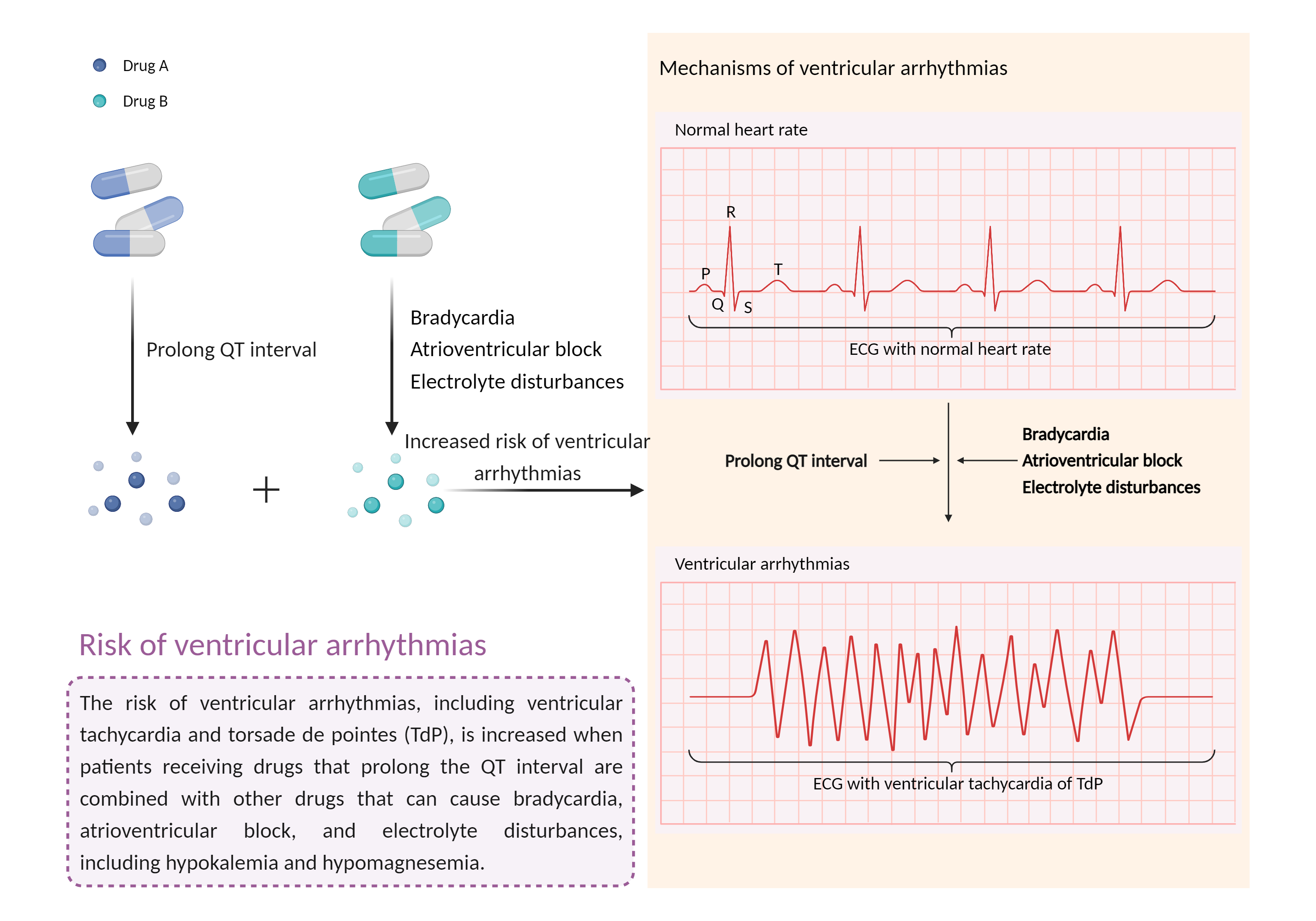 |
|||||||||
| Drug Name | Ibutilide | Orciprenaline | |||||||
| Mechanism 2 | Prolong QT interval | Hypokalemia | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Ventricular arrhythmias | ||||||||
| Factor Description | Ventricular arrhythmias are abnormal heart rhythms that cause your heart's lower chambers to pump blood instead of pumping it. This can limit or stop your heart from supplying blood to your body. While some of these arrhythmias are harmless and do not cause symptoms, others can have serious, even fatal, effects on your body. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if beta-2 adrenergic agonists are used in combination with other drugs that prolong the QT interval, including class IA and III antiarrhythmic agents, certain neuroleptic agents, phenothiazines, tricyclic antidepressants, quinolones, ketolide and macrolide antibiotics, and cisapride. It may be appropriate to monitor ECG and serum electrolytes during chronic systemic use or high-dose therapy. Patients should be advised to seek medical attention if they experience symptoms that could indicate the occurrence of torsade de pointes such as dizziness, lightheadedness, fainting, palpitations, irregular heartbeat, shortness of breath, or syncope. | ||||||||
