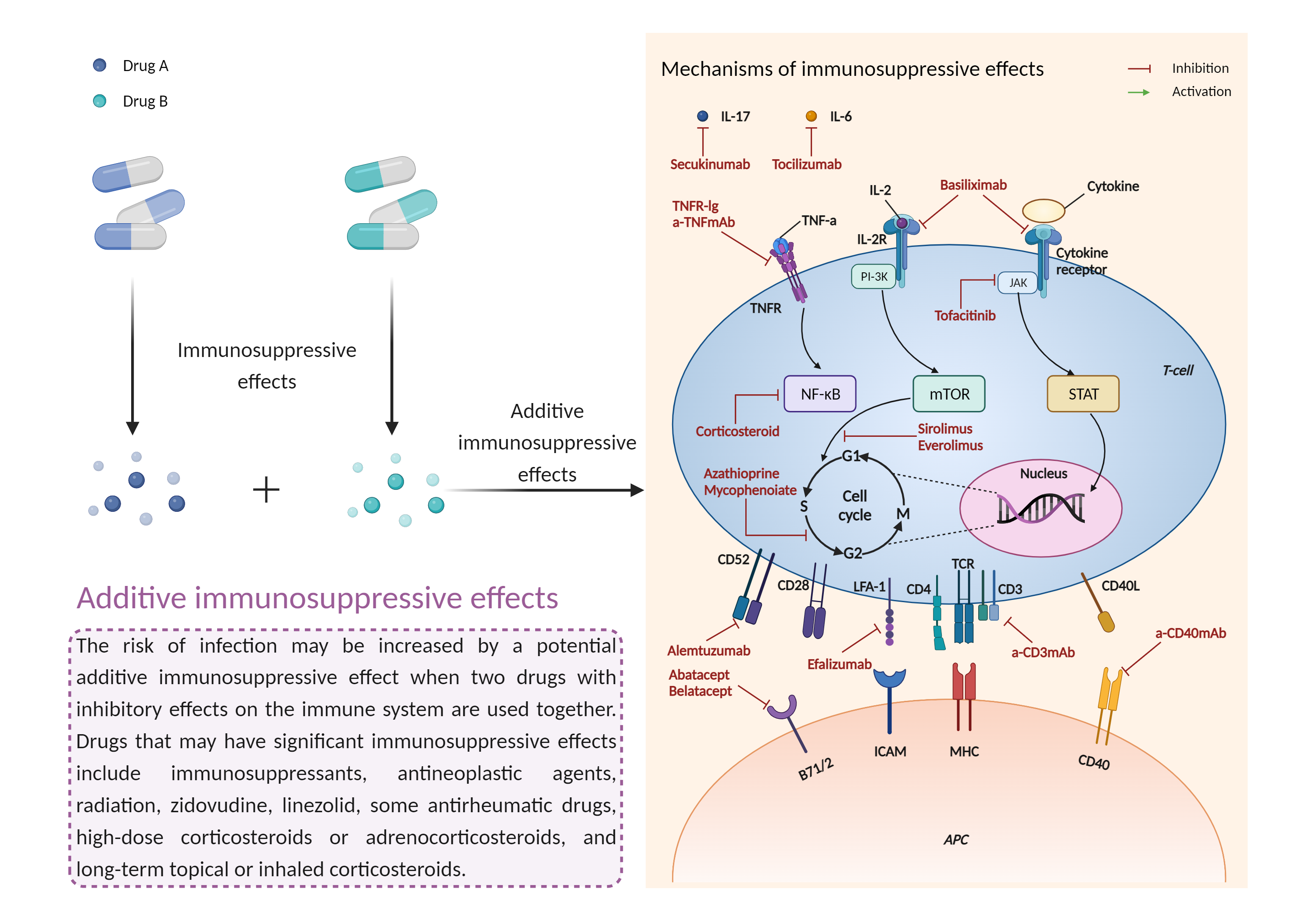
| Management |
Close monitoring for the development of infection is recommended if baricitinib or tofacitinib is used in combination with other immuno- or myelosuppressive agents, including the possible development of tuberculosis in patients who tested negative for latent tuberculosis infection prior to initiating therapy. Lymphocyte and neutrophil counts as well as hemoglobin should be evaluated at baseline and regularly during therapy, and the baricitinib or tofacitinib dosage adjusted as necessary in accordance with the product labeling. Patients should be advised to contact their physician if they develop signs and symptoms of infection such as fever, chills, diarrhea, sore throat, muscle aches, shortness of breath, blood in phlegm, weight loss, red or inflamed skin, body sores, and pain or burning during urination. If a serious infection, an opportunistic infection, or sepsis develops, baricitinib should be interrupted until the infection is controlled. Caution is recommended when using baricitinib or tofacitinib in patients with a history of diverticular disease and in patients receiving long-term concomitant treatment with drugs associated with an increased risk of DV and/or GI perforation, such as aspirin, NSAIDs, corticosteroids, and opioids. Patients should be advised to contact their healthcare provider if they experience signs of DV or GI perforation, such as severe abdominal pain, fever, nausea, or vomiting. |