Details of Drug-Drug Interaction
| Drug General Information (ID: DDIM5ERSLP) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Deferiprone | Drug Info | Trastuzumab deruxtecan | Drug Info | |||||
| Drug Type | Small molecule | Monoclonal antibody | |||||||
| Therapeutic Class | Chelating Agents | Antineoplastics/Her2 Inhibitors | |||||||
| Mechanism of Deferiprone-Trastuzumab deruxtecan Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
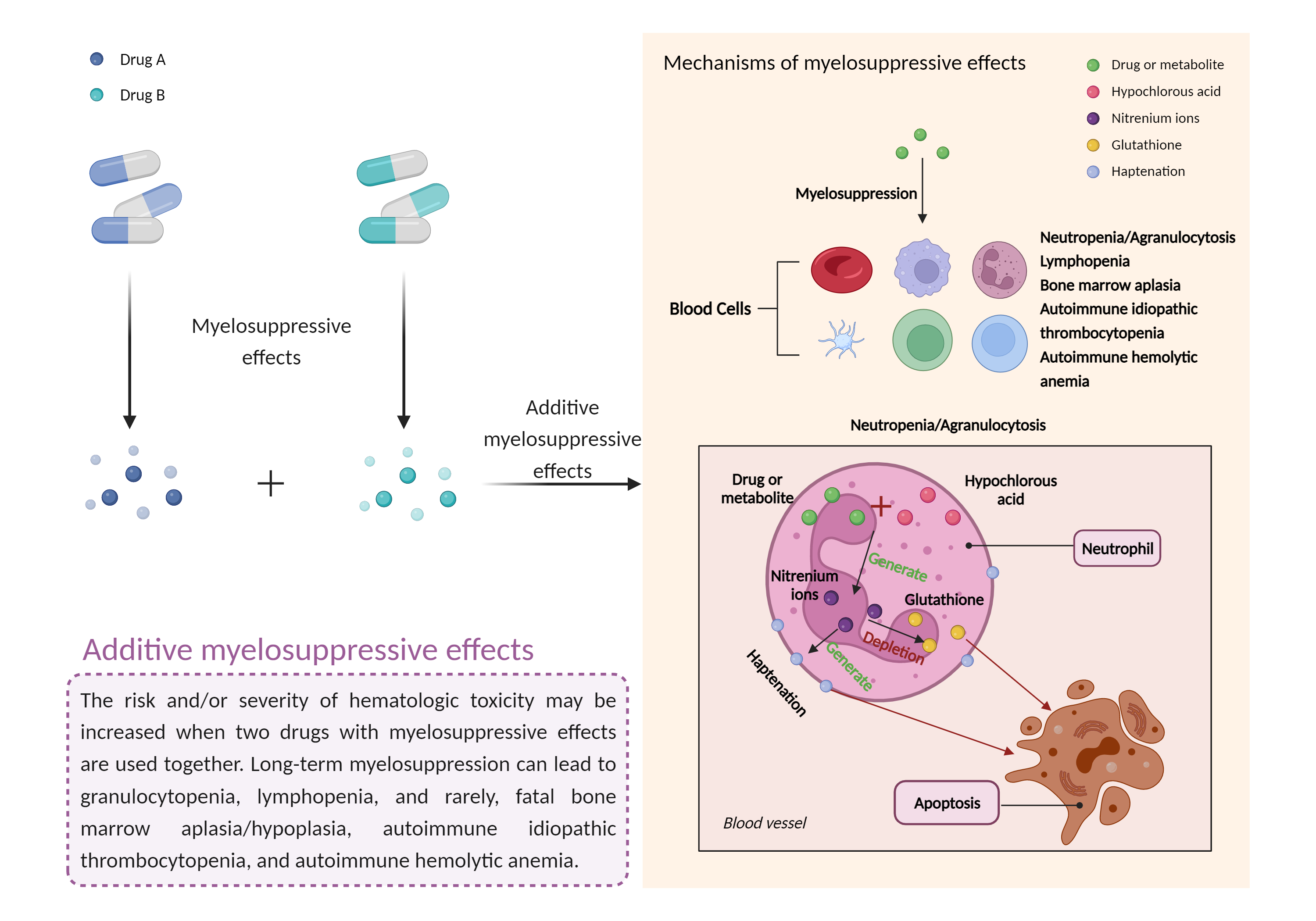
| Additive myelosuppressive effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Deferiprone | Trastuzumab deruxtecan | |||||||
| Mechanism | Myelosuppressive effects | Myelosuppressive effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Myelosuppressive effects | ||||||||
| Factor Description | Myelosuppression, also known as bone marrow suppression, is a decrease in bone marrow activity that leads to a decrease in the production of blood cells. Some blood cell disorders include: erythrocytopenia (anemia), leukopenia (neutropenia), and thrombocytopenia (thrombocytopenia). | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Concomitant use of deferiprone with other drugs known to be associated with neutropenia or agranulocytosis should generally be avoided. Otherwise, close monitoring of absolute neutrophil count (ANC) is recommended if coadministration is required. | ||||||||
