Details of Drug-Drug Interaction
| Drug General Information (ID: DDIM0FNOT4) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Interferon beta-1b | Drug Info | Bupropion | Drug Info | |||||
| Drug Type | Interferons | Small molecule | |||||||
| Therapeutic Class | Immunomodulatory Agents | Antidepressants | |||||||
| Mechanism of Interferon beta-1b-Bupropion Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
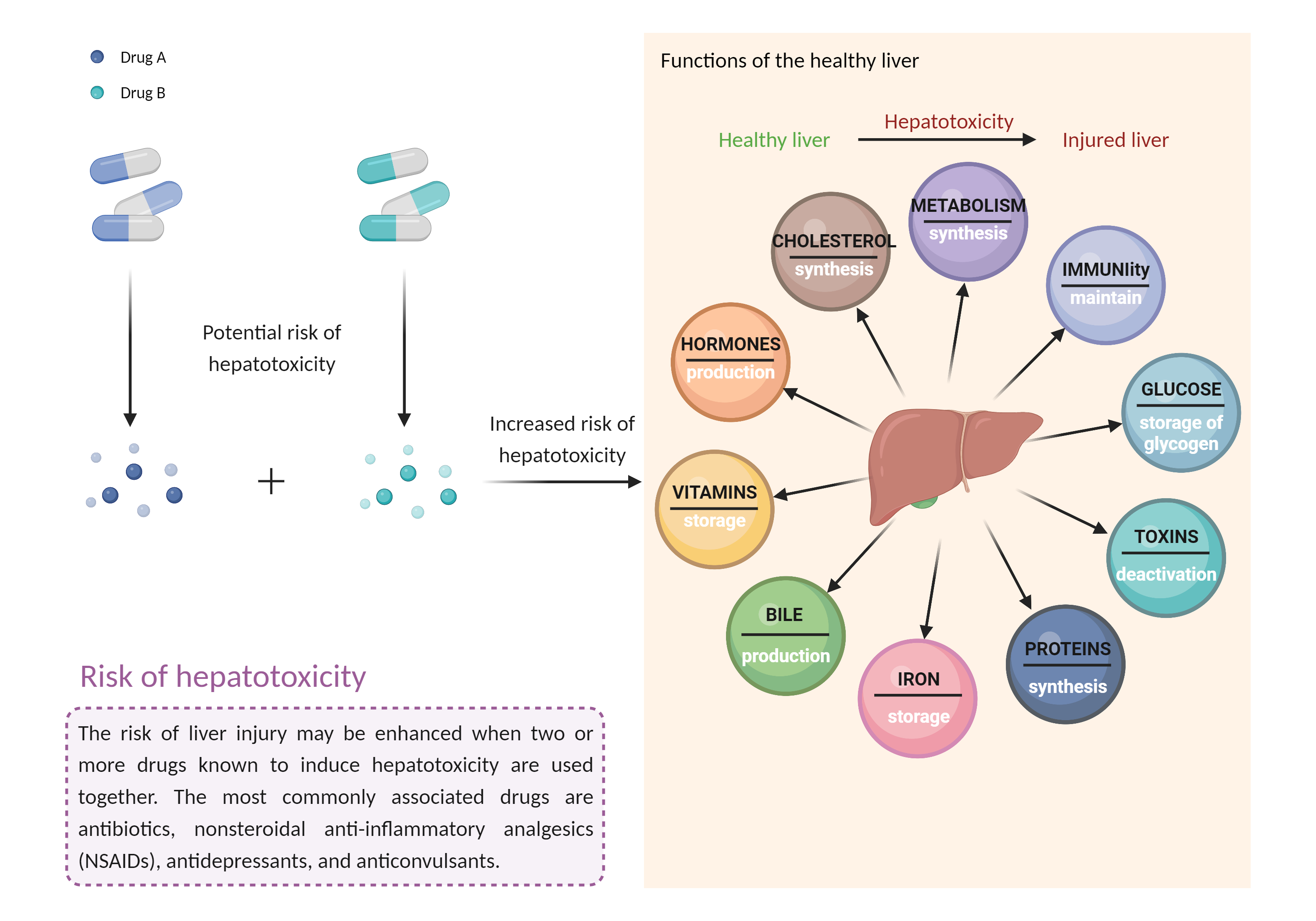
| Increased risk of hepatotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Interferon beta-1b | Bupropion | |||||||
| Mechanism 1 | Hepatotoxicity | Hepatotoxicity | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hepatotoxicity | ||||||||
| Factor Description | Combination of drugs that can induce hepatotoxicity may increase the risk of liver injury. Symptoms vary depending on the level of exposure and the total extent of liver damage, and may cause few symptoms if the damage is mild, and eventually lead to liver failure in patients with severe damage. | ||||||||
| Mechanism Description |
|
||||||||
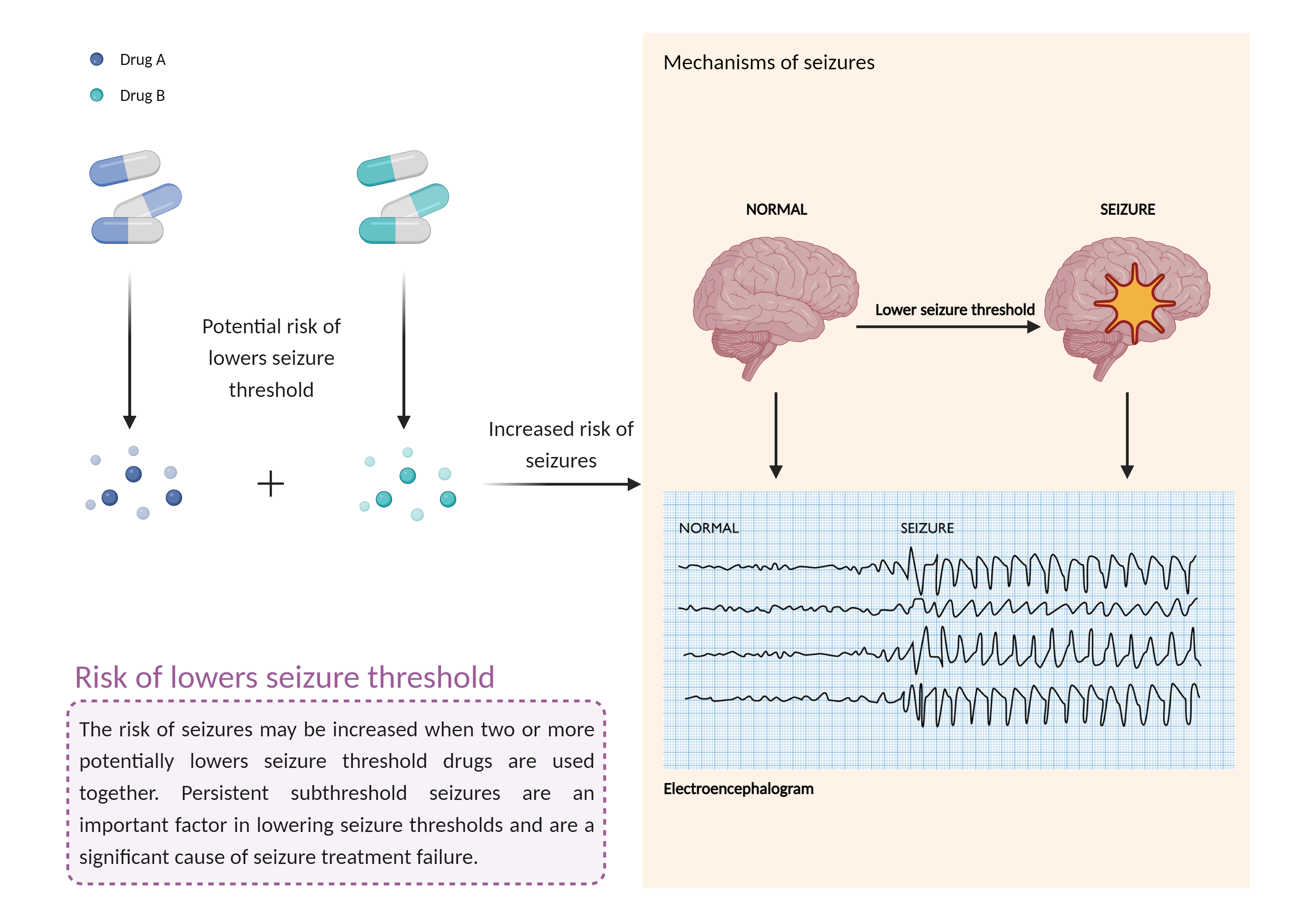
| Increased risk of lowers seizure threshold Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Interferon beta-1b | Bupropion | |||||||
| Mechanism 2 | Lower seizure threshold | Lower seizure threshold | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Lowers seizure threshold | ||||||||
| Factor Description | The combination of medications that lower the seizure threshold is a factor that makes people with epilepsy more likely to have seizures. A seizure is a sudden, uncontrolled electrical disturbance in the brain that can cause changes in your behavior, movements or sensations, and level of consciousness. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if bupropion is administered with beta interferons, particularly in the elderly and in patients with a history of seizures or other risk factors for seizures (e.g., head trauma brain tumor severe hepatic cirrhosis metabolic disorders CNS infections excessive use of alcohol or sedatives addiction to opiates, cocaine, or stimulants diabetes treated with oral hypoglycemic agents or insulin). The manufacturer recommends a lower initial dosage of bupropion and gradual dosage increments in patients receiving concurrent treatment with other potentially epileptogenic drugs. The total dose of bupropion should generally not exceed 450 mg/day (or 150 mg every other day in patients with severe hepatic cirrhosis). Bupropion should be discontinued and not restarted in patients who experience a seizure during treatment.The risk of hepatic injury should also be considered with this combination. Liver function tests should be monitored at regular intervals and the interferon dosage reduced if SGPT rises above 5 times the upper limit of normal. The dosage may be gradually re-escalated when enzyme levels return to normal. Patients should be advised to notify their physician if they experience signs and symptoms of hepatotoxicity such as fever, rash, anorexia, nausea, vomiting, fatigue, right upper quadrant pain, dark urine, and jaundice. If liver injury is suspected, interferon therapy should be promptly discontinued due to the potential for rapid progression to liver failure. | ||||||||
