Details of Drug-Drug Interaction
| Drug General Information (ID: DDIKFOX3A9) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Peginterferon alfa-2a | Drug Info | Ropinirole | Drug Info | |||||
| Drug Type | Interferons | Small molecule | |||||||
| Therapeutic Class | Antineoplastics | Antiparkinson Agents | |||||||
| Mechanism of Peginterferon alfa-2a-Ropinirole Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| CYP450 enzyme inhibition Click to Show/Hide Mechanism Graph | |||||||||
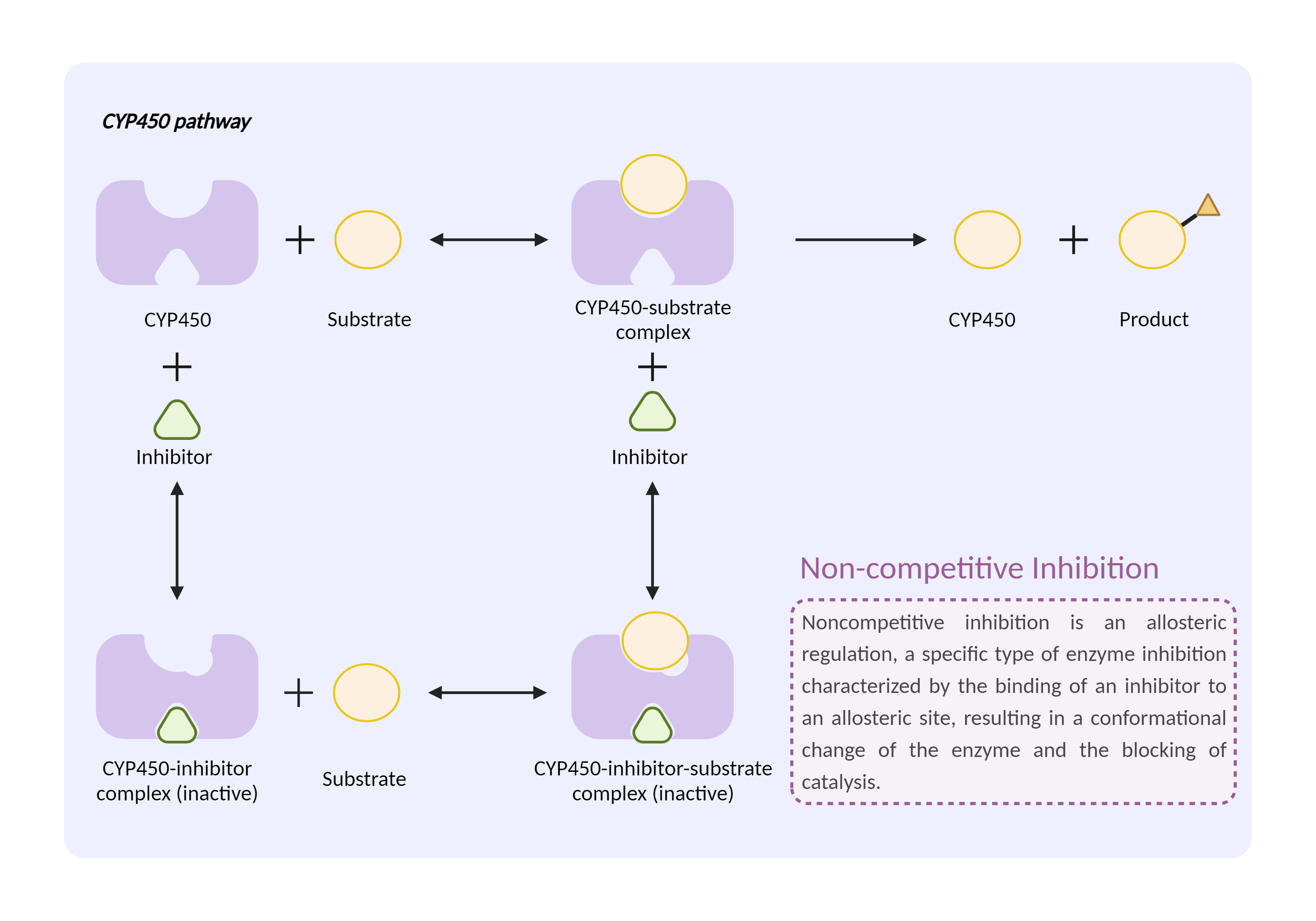 |
|||||||||
| Drug Name | Peginterferon alfa-2a | Ropinirole | |||||||
| Mechanism | CYP450 1A2 inhibitor | CYP450 1A2 substrate | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Cytochrome P450 1A2 |
×
Structure
Sequence
MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKNPHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDGQSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELMAGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFPILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGNLIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLSDRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPELWEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLEFSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
|
|||||||
| Gene Name | CYP1A2 | ||||||||
| Uniprot ID | CP1A2_HUMAN | ||||||||
| KEGG Pathway | hsa:1544 | ||||||||
| Protein Family | Cytochrome P450 family | ||||||||
| Protein Function |
A cytochrome P450 monooxygenase involved in the metabolism of various endogenous substrates, including fatty acids, steroid hormones and vitamins (PubMed:9435160, PubMed:10681376, PubMed:11555828, PubMed:12865317, PubMed:19965576). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase) (PubMed:9435160, PubMed:10681376, PubMed:11555828, PubMed:12865317, PubMed:19965576). Catalyzes the hydroxylation of carbon-hydrogen bonds (PubMed:11555828, PubMed:12865317). Exhibits high catalytic activity for the formation of hydroxyestrogens from estrone (E1) and 17beta-estradiol (E2), namely 2-hydroxy E1 and E2 (PubMed:11555828, PubMed:12865317). Metabolizes cholesterol toward 25-hydroxycholesterol, a physiological regulator of cellular cholesterol homeostasis (PubMed:21576599). May act as a major enzyme for all-trans retinoic acid biosynthesis in the liver. Catalyzes two successive oxidative transformation of all-trans retinol to all-trans retinal and then to the active form all-trans retinoic acid (PubMed:10681376). Primarily catalyzes stereoselective epoxidation of the last double bond of polyunsaturated fatty acids (PUFA), displaying a strong preference for the (R,S) stereoisomer (PubMed:19965576). Catalyzes bisallylic hydroxylation and omega-1 hydroxylation of PUFA (PubMed:9435160). May also participate in eicosanoids metabolism by converting hydroperoxide species into oxo metabolites (lipoxygenase-like reaction, NADPH-independent) (PubMed:21068195). Plays a role in the oxidative metabolism of xenobiotics. Catalyzes the N-hydroxylation of heterocyclic amines and the O-deethylation of phenacetin (PubMed:14725854). Metabolizes caffeine via N3-demethylation (Probable).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Pharmacologic response to ropinirole should be monitored more closely whenever a CYP450 1A2 inhibitor is added to or withdrawn from therapy, and the ropinirole dosage adjusted as necessary. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Noroxin (norfloxacin). Merck & Co, Inc, West Point, PA. | ||||||||||||||||||
| 2 | Product Information. Requip (ropinirole). SmithKline Beecham, Philadelphia, PA. | ||||||||||||||||||
