Details of Drug-Drug Interaction
| Drug General Information (ID: DDIK0XRBT1) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Rituximab | Drug Info | Baricitinib | Drug Info | |||||
| Drug Type | Monoclonal antibody | Small molecule | |||||||
| Therapeutic Class | Antineoplastics | Antirheumatics | |||||||
| Mechanism of Rituximab-Baricitinib Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Additive immunosuppressive effects Click to Show/Hide Mechanism Graph | |||||||||
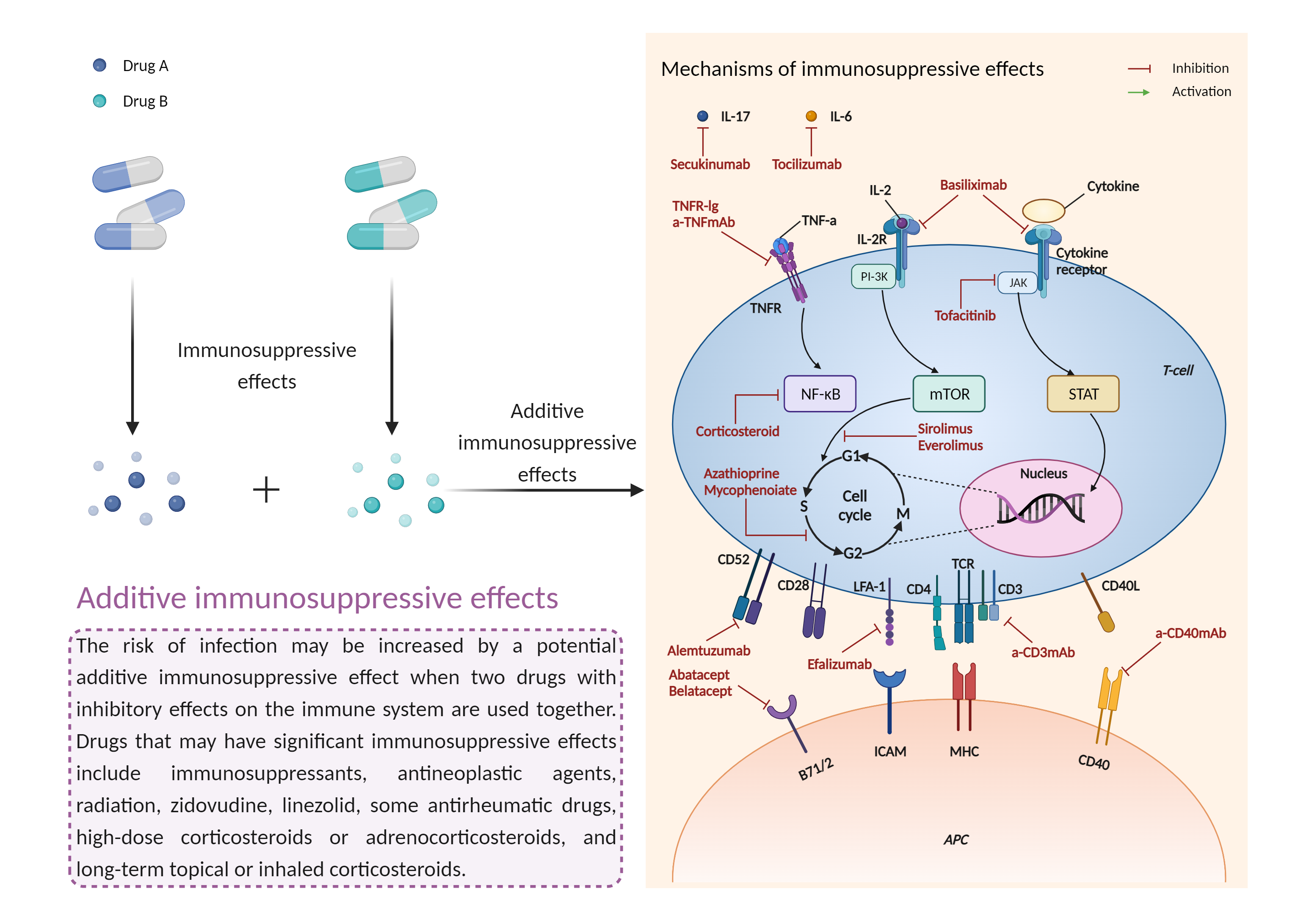 |
|||||||||
| Drug Name | Rituximab | Baricitinib | |||||||
| Mechanism | Immunosuppressive effects | Immunosuppressive effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Immunosuppressive effects | ||||||||
| Factor Description | Immunosuppression is when your immune system is not functioning as it should. The immune system is made up of cells, tissues and organs that help the body fight off infections. If the immune system is suppressed, an infection that your body was able to control may become serious or even fatal. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Concomitant use of baricitinib with other Janus kinase (JAK) inhibitors, biologic disease-modifying antirheumatic drugs (DMARDs), or potent immunosuppressants (e.g., azathioprine, cyclosporine) is not recommended. Patients receiving baricitinib should be closely monitored for the development of signs and symptoms of infection during and after treatment, including the possible development of tuberculosis in patients who tested negative for latent tuberculosis infection prior to initiating therapy. If a serious infection, an opportunistic infection, or sepsis develops, baricitinib should be interrupted until the infection is controlled. | ||||||||
