Details of Drug-Drug Interaction
| Drug General Information (ID: DDIJMOWXQT) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Tolbutamide | Drug Info | Dabrafenib | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antidiabetic Agents | Multikinase Inhibitors | |||||||
| Structure | |||||||||
| Mechanism of Tolbutamide-Dabrafenib Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| CYP450 enzyme induction Click to Show/Hide Mechanism Graph | |||||||||
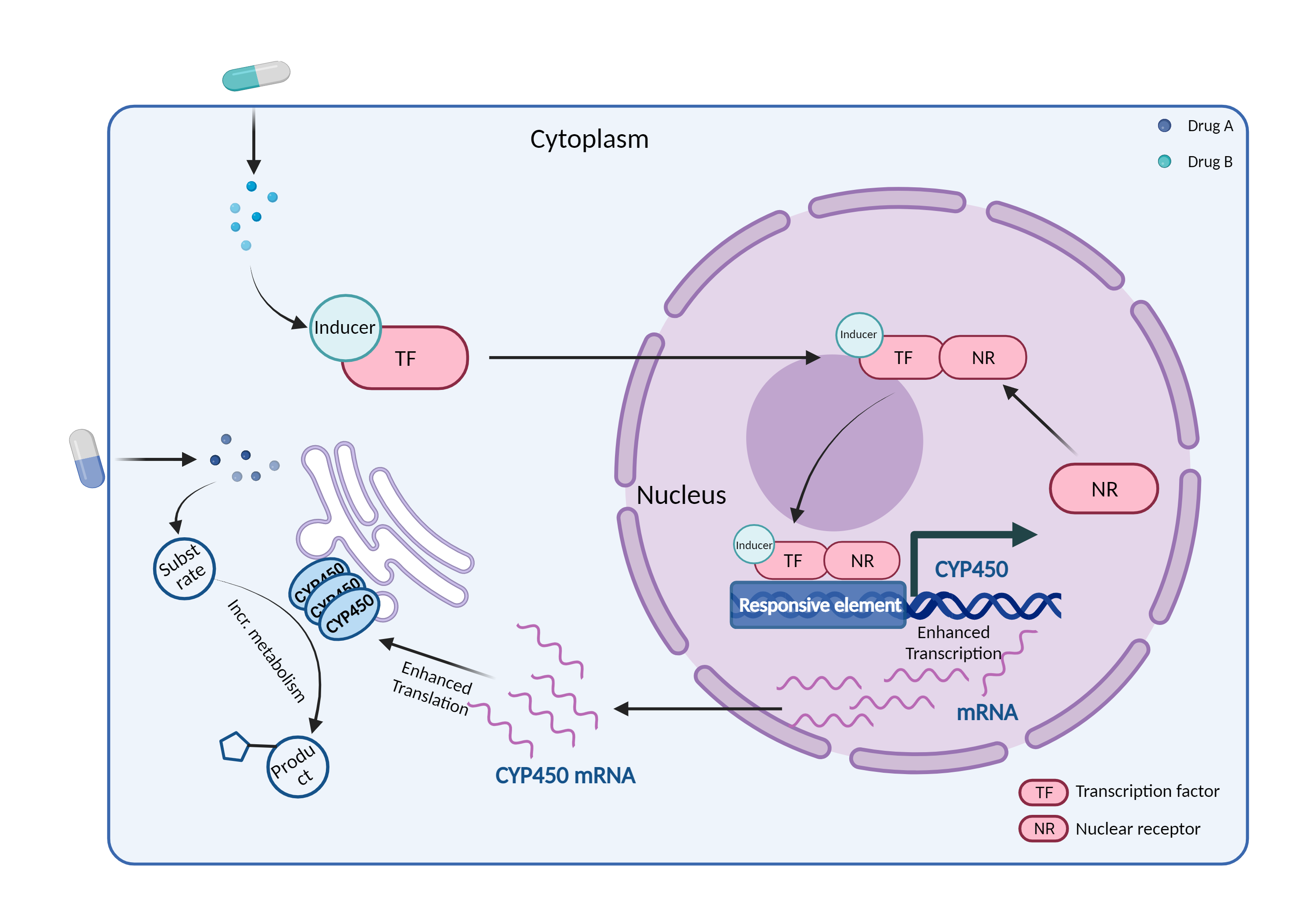 |
|||||||||
| Drug Name | Tolbutamide | Dabrafenib | |||||||
| Mechanism | CYP450 2C9 substrate | CYP450 2C9 inducer | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Cytochrome P450 2C9 |
×
Structure
Sequence
MDSLVVLVLCLSCLLLLSLWRQSSGRGKLPPGPTPLPVIGNILQIGIKDISKSLTNLSKVYGPVFTLYFGLKPIVVLHGYEAVKEALIDLGEEFSGRGIFPLAERANRGFGIVFSNGKKWKEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICSIIFHKRFDYKDQQFLNLMEKLNENIKILSSPWIQICNNFSPIIDYFPGTHNKLLKNVAFMKSYILEKVKEHQESMDMNNPQDFIDCFLMKMEKEKHNQPSEFTIESLENTAVDLFGAGTETTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRSHMPYTDAVVHEVQRYIDLLPTSLPHAVTCDIKFRNYLIPKGTTILISLTSVLHDNKEFPNPEMFDPHHFLDEGGNFKKSKYFMPFSAGKRICVGEALAGMELFLFLTSILQNFNLKSLVDPKNLDTTPVVNGFASVPPFYQLCFIPV
|
|||||||
| Gene Name | CYP2C9 | ||||||||
| Uniprot ID | CP2C9_HUMAN | ||||||||
| KEGG Pathway | hsa:1559 | ||||||||
| Protein Family | Cytochrome P450 family | ||||||||
| Protein Function |
A cytochrome P450 monooxygenase involved in the metabolism of various endogenous substrates, including fatty acids and steroids (PubMed:7574697, PubMed:9866708, PubMed:9435160, PubMed:12865317, PubMed:15766564, PubMed:19965576, PubMed:21576599). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase) (PubMed:7574697, PubMed:9866708, PubMed:9435160, PubMed:12865317, PubMed:15766564, PubMed:19965576, PubMed:21576599). Catalyzes the epoxidation of double bonds of polyunsaturated fatty acids (PUFA) (PubMed:7574697, PubMed:15766564, PubMed:19965576, PubMed:9866708). Catalyzes the hydroxylation of carbon-hydrogen bonds. Metabolizes cholesterol toward 25-hydroxycholesterol, a physiological regulator of cellular cholesterol homeostasis (PubMed:21576599). Exhibits low catalytic activity for the formation of catechol estrogens from 17beta-estradiol (E2) and estrone (E1), namely 2-hydroxy E1 and E2 (PubMed:12865317). Catalyzes bisallylic hydroxylation and hydroxylation with double-bond migration of polyunsaturated fatty acids (PUFA) (PubMed:9866708, PubMed:9435160). Also metabolizes plant monoterpenes such as limonene. Oxygenates (R)- and (S)-limonene to produce carveol and perillyl alcohol (PubMed:11950794). Contributes to the wide pharmacokinetics variability of the metabolism of drugs such as S-warfarin, diclofenac, phenytoin, tolbutamide and losartan (PubMed:25994031).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised when dabrafenib is prescribed to patients with diabetes, particularly those receiving antidiabetic agents that are metabolized by CYP450 3A4 and/or 2C9. Close clinical monitoring of glycemic control is recommended following initiation or discontinuation of dabrafenib, and the dosages of concomitant antidiabetic agents adjusted as necessary. Patients should be advised to notify their physician if their blood glucose is consistently high or if they experience symptoms of severe hyperglycemia such as excessive thirst and increases in the volume or frequency of urination. Likewise, patients should be observed for hypoglycemia when dabrafenib is withdrawn from their therapeutic regimen. | ||||||||
