Details of Drug-Drug Interaction
| Drug General Information (ID: DDIIZQY769) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Digitoxin | Drug Info | Tofacitinib | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiarrhythmic Agents | Antirheumatics | |||||||
| Structure | |||||||||
| Mechanism of Digitoxin-Tofacitinib Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
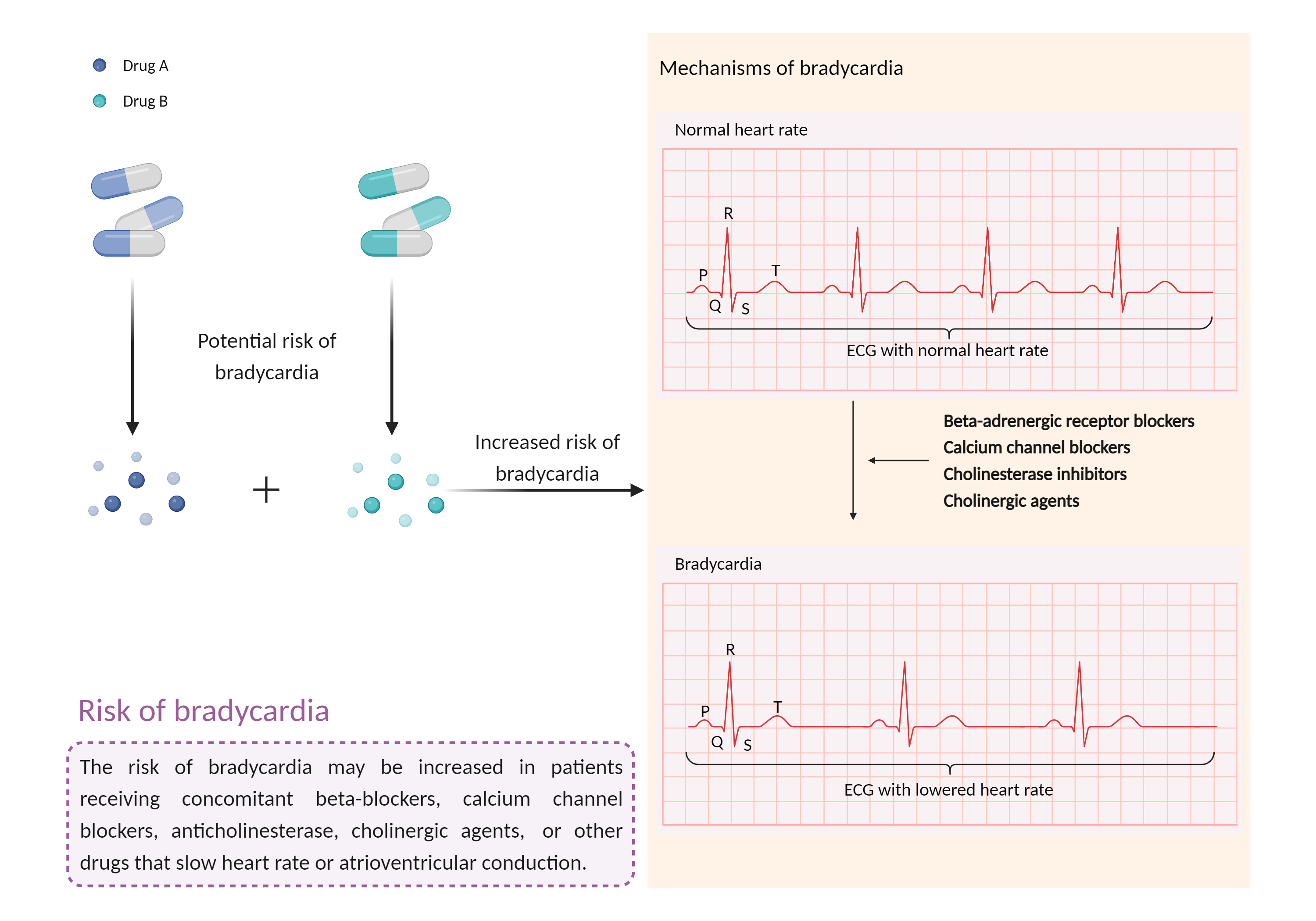
| Increased risk of bradycardia Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Digitoxin | Tofacitinib | |||||||
| Mechanism | Bradycardia | Bradycardia | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Bradycardia | ||||||||
| Factor Description | Bradycardia is a slow heart rate in which the heart beats less than 60 times per minute. If the heart rate is very slow and the heart is not pumping enough oxygen-rich blood to the body, and you may feel dizzy, very tired or weak, and short of breath. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | According to the manufacturer, concomitant use of tofacitinib with other agents that lower heart rate and/or prolong the PR interval should be avoided when possible. Caution is advised if concomitant use is necessary, particularly in patients with known conduction problems or severe cardiac disease. An ECG should be considered in these patients before initiating concomitant therapy and after titration to steady-state. Patients should be advised to notify their physician if they experience dizziness, lightheadedness, fainting, or irregular heartbeats. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Cerner Multum, Inc. "Australian Product Information.". | ||||||||||||||||||
| 2 | Canadian Pharmacists Association "e-CPS." | ||||||||||||||||||
