Details of Drug-Drug Interaction
| Drug General Information (ID: DDIIZ4563D) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Alfuzosin | Drug Info | Paliperidone | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antihypertensive Agents | Atypical Antipsychotics | |||||||
| Structure | |||||||||
| Mechanism of Alfuzosin-Paliperidone Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Additive hypotensive effects Click to Show/Hide Mechanism Graph | |||||||||
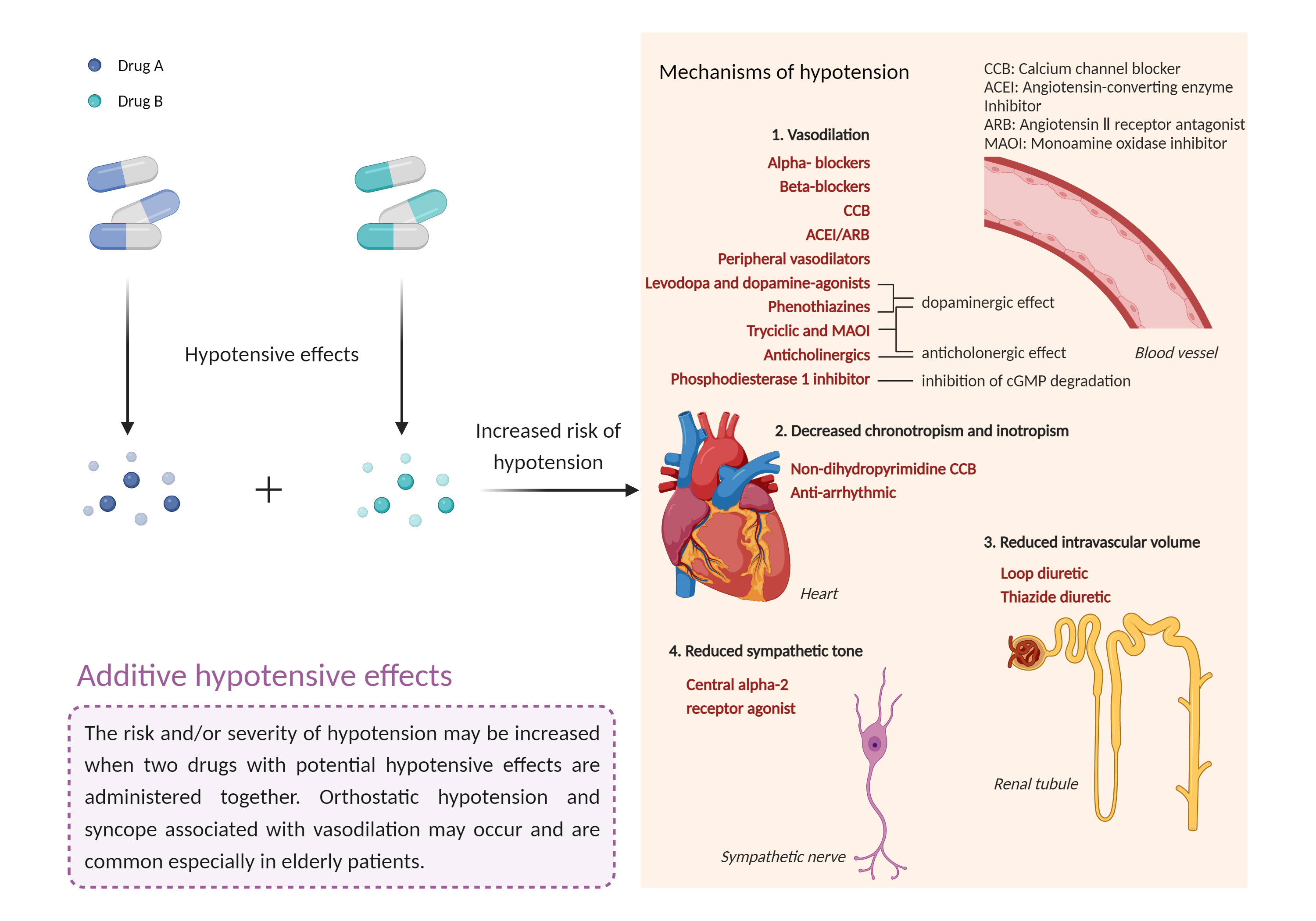 |
|||||||||
| Drug Name | Alfuzosin | Paliperidone | |||||||
| Mechanism 1 |
Hypotensive effects Alpha-1 adrenergic receptor Antagonist |
Hypotensive effects Alpha-1 adrenergic receptor Antagonist |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Adrenergic receptor alpha-1 | Structure Sequence | |||||||
| Protein Family | G-protein coupled receptor 1 family | ||||||||
| Protein Function |
This alpha-adrenergic receptor mediates its action by association with G proteins that activate a phosphatidylinositol-calcium second messenger system. Its effect is mediated by G(q) and G(11) proteins. Nuclear ADRA1A-ADRA1B heterooligomers regulate phenylephrine(PE)-stimulated ERK signaling in cardiac myocytes.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Increased risk of prolong QT interval Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Alfuzosin | Paliperidone | |||||||
| Mechanism 2 | Prolong QT interval | Prolong QT interval | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | QT interval | ||||||||
| Factor Description | Long QT syndrome is a heart signaling disorder that can cause a fast, chaotic heartbeat (arrhythmia). Many people may not exhibit symptoms, and usually the condition is detected during routine medical tests. In others, the most common symptoms include: sudden fainting, palpitations, dizziness, seizures, sudden death. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Close clinical monitoring for adverse effects is recommended during concurrent therapy. A lower starting dosage and slower titration of the neuroleptic agent may be appropriate in patients receiving alfuzosin, especially if they are elderly. Patients should be advised to notify their physician if they experience orthostatic symptoms (dizziness, lightheadedness, syncope), and to seek medical help if they experience palpitations and fast/ or irregular heartbeats. | ||||||||
