Details of Drug-Drug Interaction
| Drug General Information (ID: DDIIXQVPWJ) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Paclitaxel (protein-bound) | Drug Info | Smallpox (Vaccinia) Vaccine, Live | Drug Info | |||||
| Drug Type | Small molecule | Vaccine | |||||||
| Therapeutic Class | Antineoplastics/Mitotic Inhibitors | Vaccine | |||||||
| Mechanism of Paclitaxel (protein-bound)-Smallpox (Vaccinia) Vaccine, Live Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
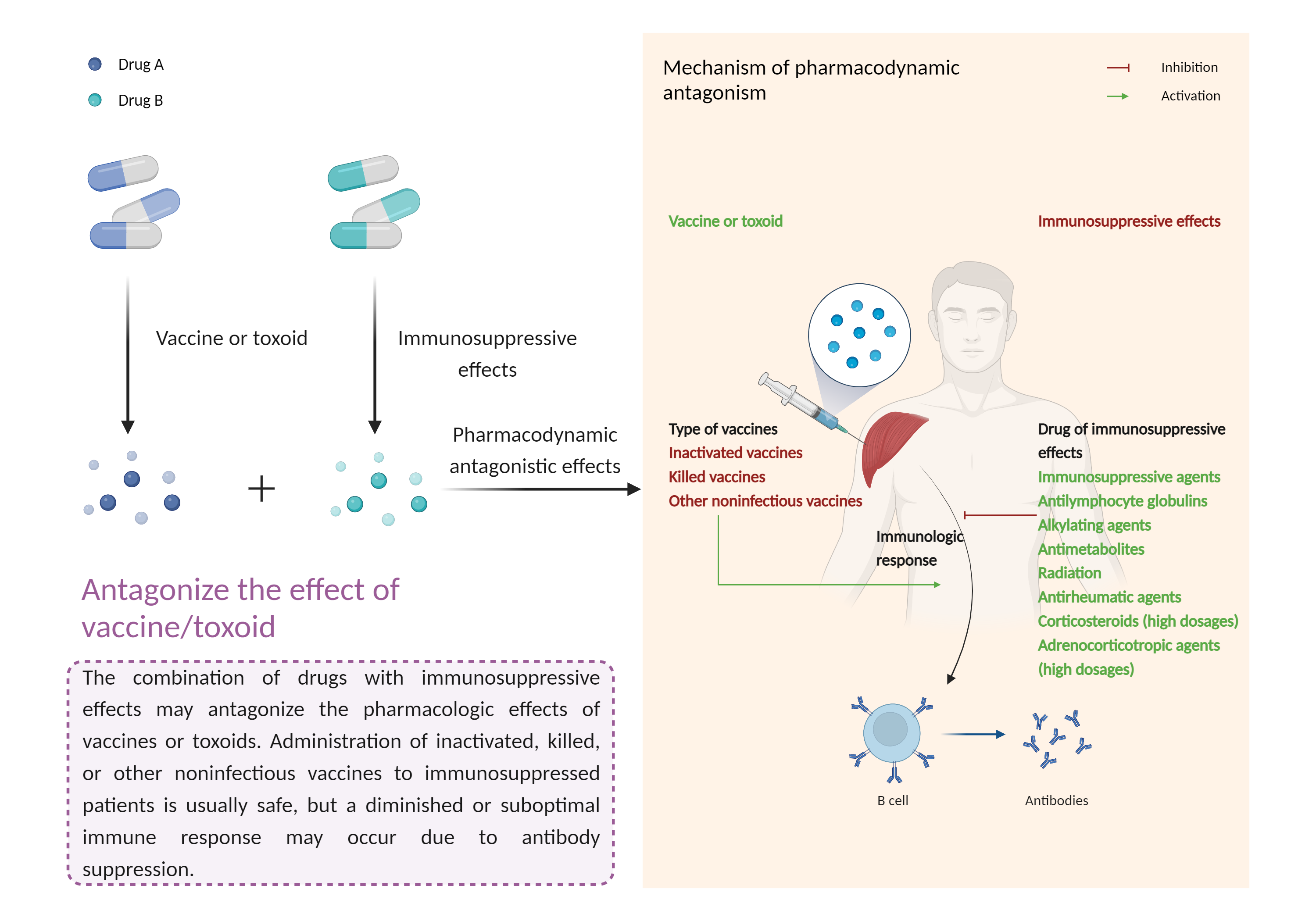
| Antagonize the effect of vaccine/toxoid Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Paclitaxel (protein-bound) | Smallpox (Vaccinia) Vaccine, Live | |||||||
| Mechanism | Immunosuppressive effects | Vaccine or toxoid | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Vaccine/toxoid | ||||||||
| Factor Description | The beneficial effects of the vaccine/toxinoid may be reduced, leading to a diminished or suboptimal immune response. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Routine, nonemergency smallpox vaccination is contraindicated in patients receiving immunosuppressive therapy or cancer chemotherapy. Vaccination should be deferred until after such therapy is discontinued for at least 3 months in most cases. A longer waiting period may be necessary following treatment with agents that have a prolonged elimination half-life (e.g., leflunomide, teriflunomide). | ||||||||
