Details of Drug-Drug Interaction
| Drug General Information (ID: DDIHLN8QY4) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Pyridostigmine | Drug Info | Prednisone | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Parasympathomimetics | Antiinflammatory Agents | |||||||
| Structure | |||||||||
| Mechanism of Pyridostigmine-Prednisone Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
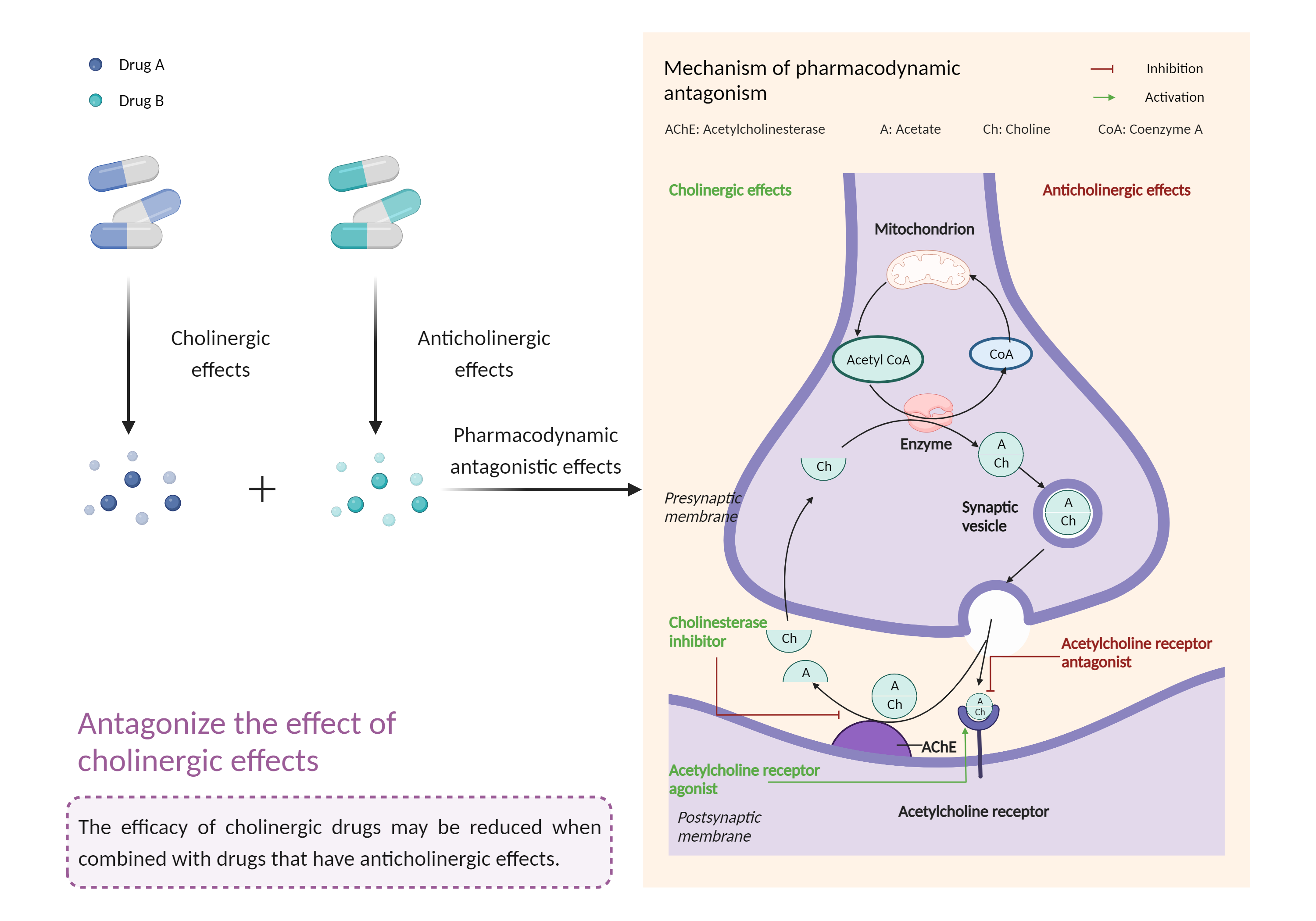
| Antagonize the effect of cholinergic agents Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Pyridostigmine | Prednisone | |||||||
| Mechanism | Acetylcholinesterase inhibitor | Reduce the therapeutic effects of acetylcholinesterase inhibitor in myasthenia gravis | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Cholinergic agents | ||||||||
| Factor Description | The beneficial effects of cholinergic drugs may be reduced, such as reduced efficacy in the treatment of Alzheimer's disease and exacerbation of symptoms in the treatment of myasthenia gravis. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Corticosteroid therapy should be instituted at relatively low dosages (15 to 25 mg/day of prednisone or equivalent) and in a controlled setting in patients with myasthenia gravis. Respiratory support should be available, and the dosage should be increased stepwise as tolerated (approximately 5 mg/day of prednisone or equivalent at 2- to 3-day intervals until marked clinical improvement or a dosage of 50 mg/day is reached). Dose reductions of the acetylcholinesterase inhibitor may be required as symptoms improve, which often may be delayed and gradual. | ||||||||
