Details of Drug-Drug Interaction
| Drug General Information (ID: DDIHF4LC6T) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Rubella virus vaccine | Drug Info | Ocrelizumab | Drug Info | |||||
| Drug Type | Vaccine | Monoclonal antibody | |||||||
| Therapeutic Class | Vaccine | Antineoplastics | |||||||
| Mechanism of Rubella virus vaccine-Ocrelizumab Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Antagonize the effect of vaccine/toxoid Click to Show/Hide Mechanism Graph | |||||||||
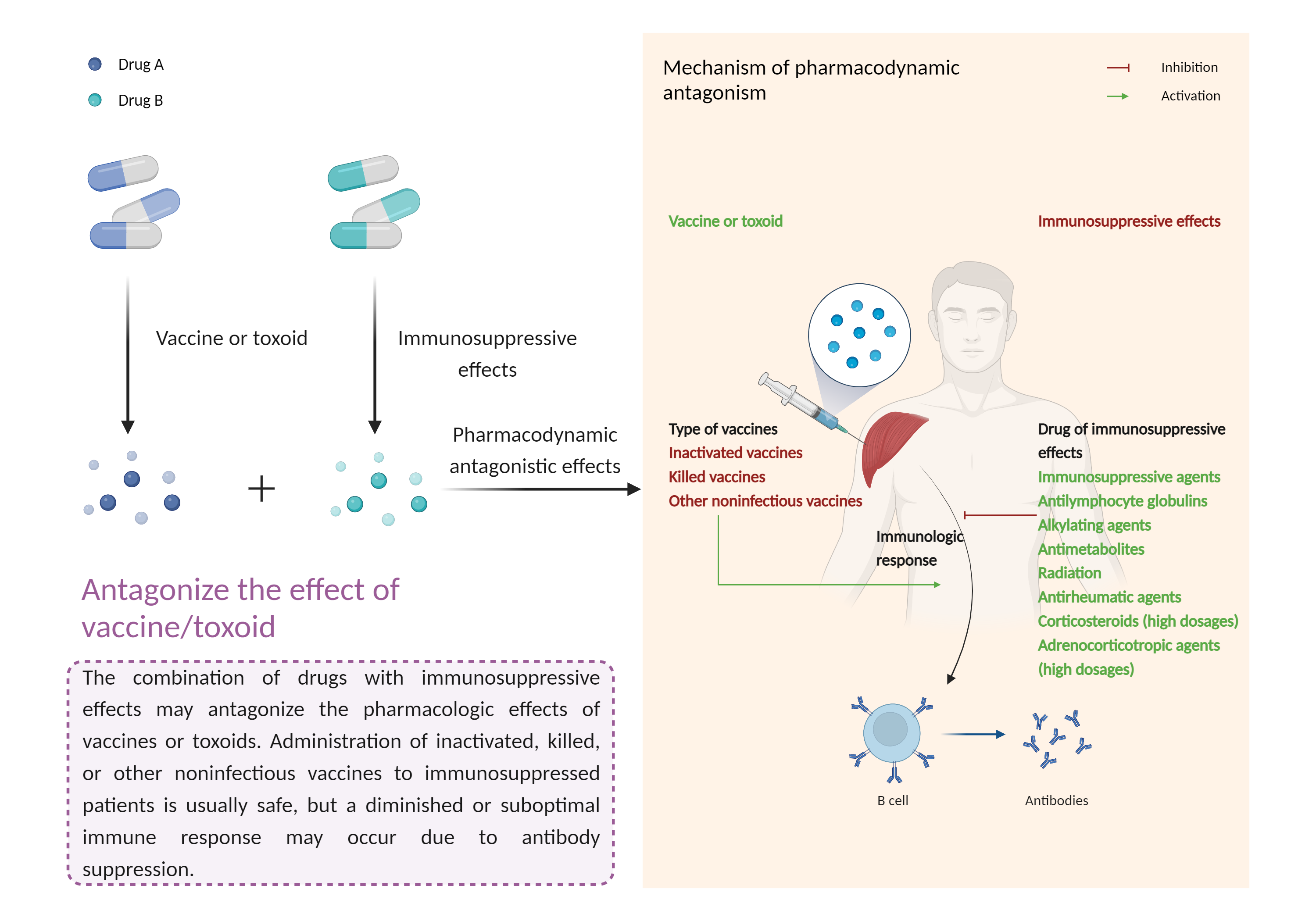 |
|||||||||
| Drug Name | Rubella virus vaccine | Ocrelizumab | |||||||
| Mechanism | Vaccine or toxoid | Immunosuppressive effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Vaccine/toxoid | ||||||||
| Factor Description | The beneficial effects of the vaccine/toxinoid may be reduced, leading to a diminished or suboptimal immune response. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Live virus or bacterial vaccines should not be administered to patients during therapy or after discontinuing ocrelizumab therapy until recovery from B-cell repletion occurs. Immunization status should be assessed prior to initiating ocrelizumab and recommended immunization with live or attenuated live vaccines should be completed at least 4 weeks prior to ocrelizumab initiation (some authorities have recommended at least 6 weeks). For infants of mothers exposed to ocrelizumab during pregnancy, live or live-attenuated vaccines should be delayed until B-cell levels have recovered therefore, measuring CD19 positive B-cell levels prior to vaccination in these infants is recommended. | ||||||||
