| Mechanism of Edrophonium-Fingolimod Interaction
(Severity Level: Moderate)
|
|
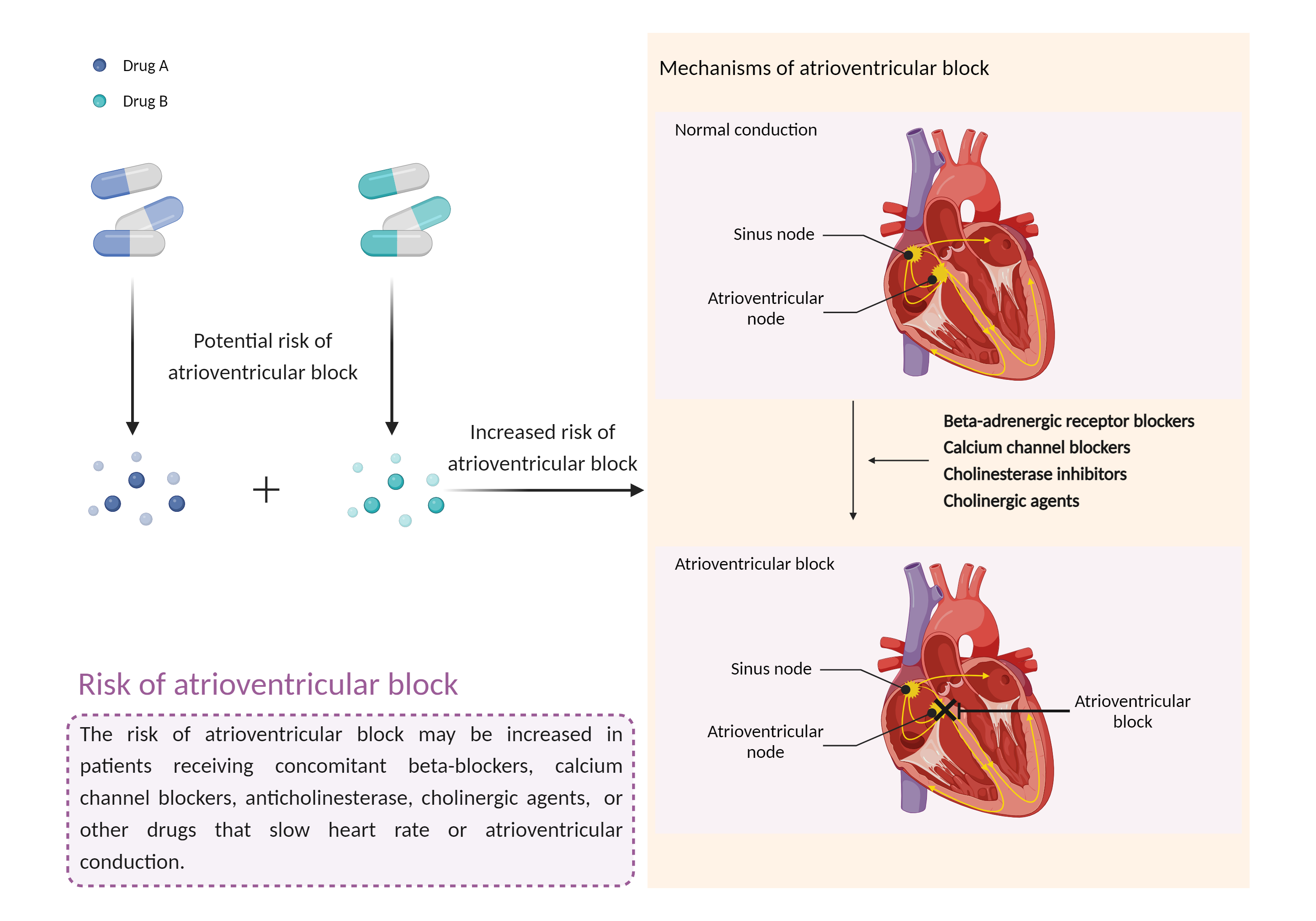
Increased risk of atrioventricular block
Click to Show/Hide Mechanism Graph
|
 |
| Drug Name |
Edrophonium |
Fingolimod |
|
Mechanism 1
|
Delay atrioventricular conduction |
Delay atrioventricular conduction |
| Key Mechanism Factor 1 |
| Factor Name |
Atrioventricular block |
| Factor Description |
Atrioventricular block is a type of cardiac conduction block that occurs when the electrical signal from the atria to the ventricles is impaired. In an Atrioventricular block, this electrical signal is either delayed or completely blocked. When the signal is completely blocked, the ventricles produce their own electrical signal to control the heart rate. The heart rate produced by the ventricles is much slower than that produced by the sinus node. |
| Mechanism Description |
- Increased risk of atrioventricular block by the combination of Edrophonium and Fingolimod
|
|
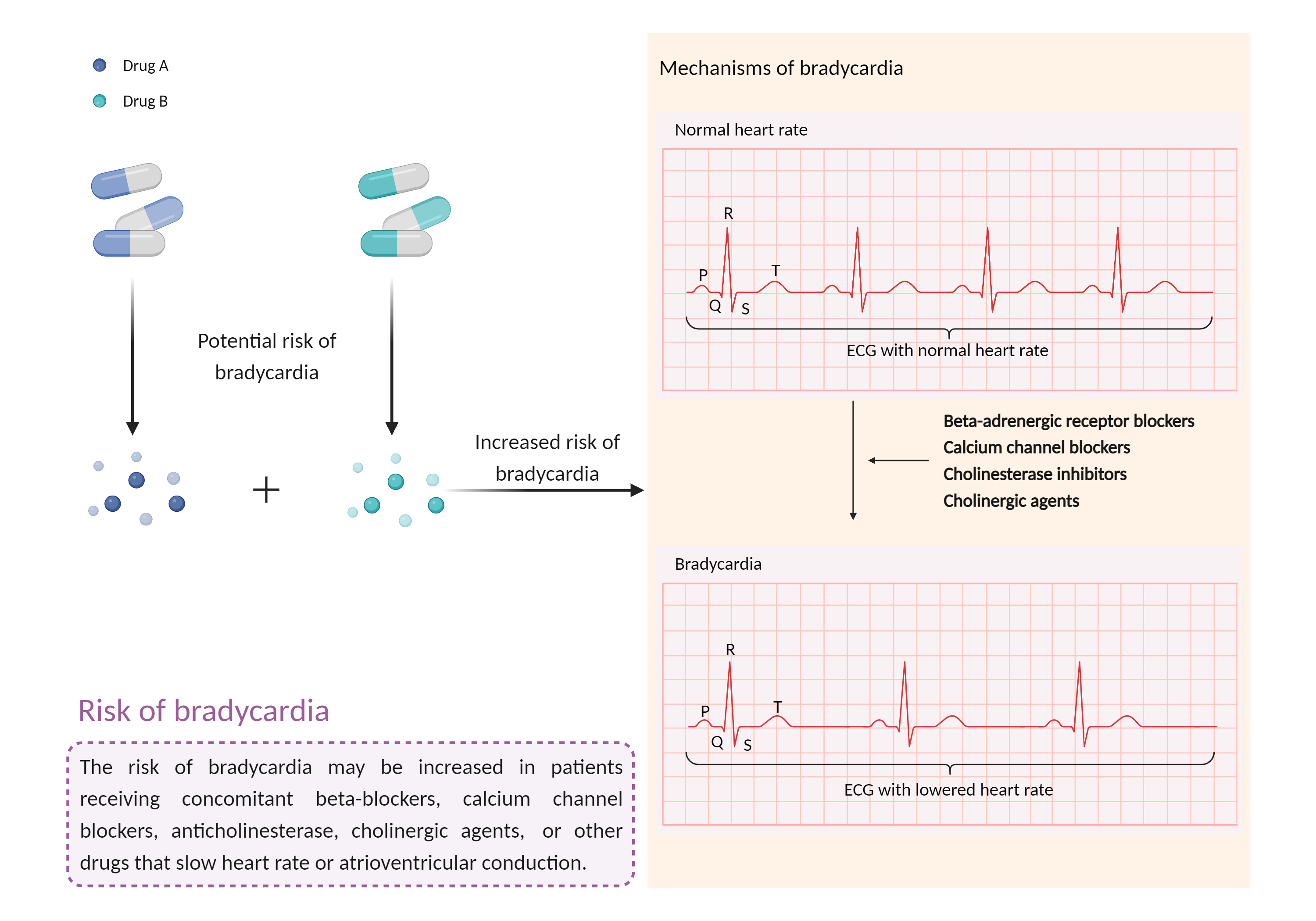
Increased risk of bradycardia
Click to Show/Hide Mechanism Graph
|
 |
| Drug Name |
Edrophonium |
Fingolimod |
|
Mechanism 2
|
Bradycardia |
Bradycardia |
| Key Mechanism Factor 2 |
| Factor Name |
Bradycardia |
| Factor Description |
Bradycardia is a slow heart rate in which the heart beats less than 60 times per minute. If the heart rate is very slow and the heart is not pumping enough oxygen-rich blood to the body, and you may feel dizzy, very tired or weak, and short of breath. |
| Mechanism Description |
- Increased risk of bradycardia by the combination of Edrophonium and Fingolimod
|


