Details of Drug-Drug Interaction
| Drug General Information (ID: DDIFX4JTVH) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Benzatropine | Drug Info | Potassium chloride | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiparkinson Agents | Dietary Supplement | |||||||
| Structure | |||||||||
| Mechanism of Benzatropine-Potassium chloride Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
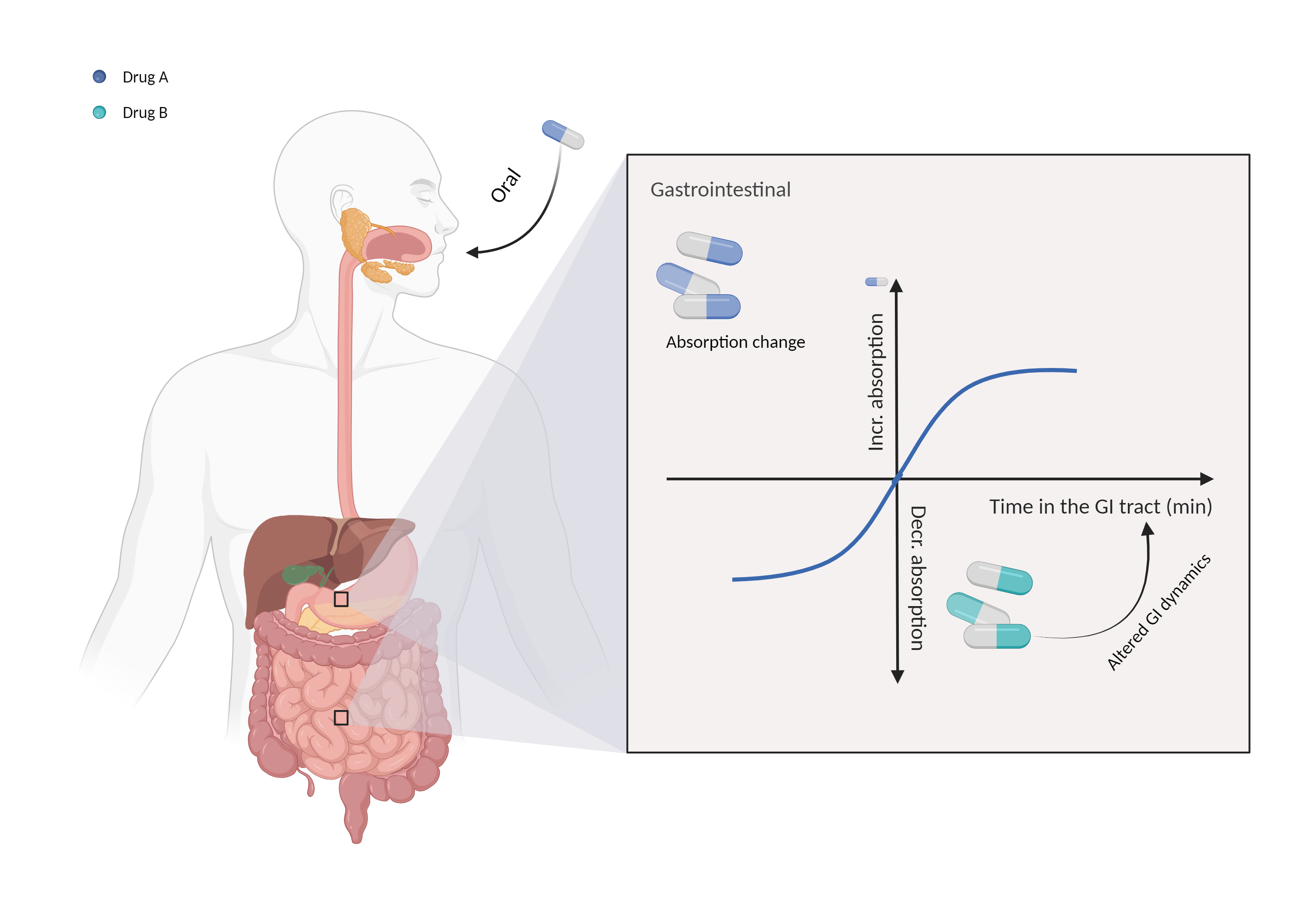
| Altered gastrointestinal dynamics Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Benzatropine | Potassium chloride | |||||||
| Mechanism | Delayed gastric emptying | Absorption affected by delayed gastric emptying | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Gastrointestinal Motility | ||||||||
| Factor Description | Gastrointestinal motility is an important factor in determining the absorption of orally administered drugs, and it controls the residence time of the drug in the digestive tract. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The use of oral solid formulations of potassium chloride is considered contraindicated in patients receiving agents with anticholinergic properties at sufficient doses to exert anticholinergic effects. A liquid formulation of potassium chloride should be considered. Patients prescribed a solid oral formulation should be advised to discontinue potassium therapy and contact their physician if they experience potential symptoms of upper GI injury such as severe vomiting, abdominal pain, distention, and gastrointestinal bleeding. | ||||||||
