Details of Drug-Drug Interaction
| Drug General Information (ID: DDIFURZPYL) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Prazosin | Drug Info | Nabumetone | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antihypertensive Agents | Nsaids/Analgesics | |||||||
| Structure | |||||||||
| Mechanism of Prazosin-Nabumetone Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Antagonize the effect of antihypertensive agents Click to Show/Hide Mechanism Graph | |||||||||
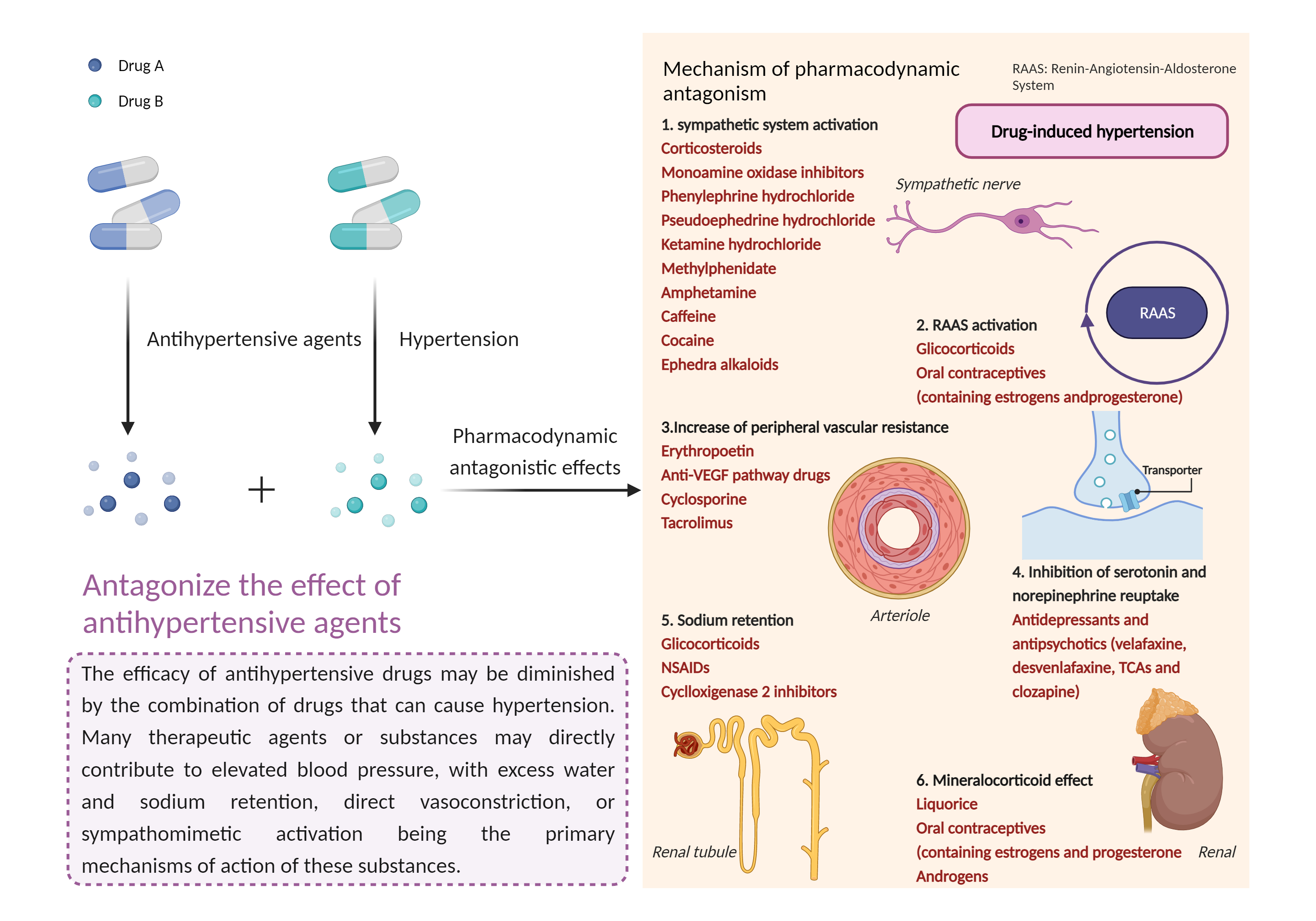 |
|||||||||
| Drug Name | Prazosin | Nabumetone | |||||||
| Mechanism |
Antihypertensive agent Alpha-1 adrenergic receptor Antagonist |
Hypertensive effects Prostaglandin G/H synthase Inhibitor |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Adrenergic receptor alpha-1 | Structure Sequence | |||||||
| Protein Family | G-protein coupled receptor 1 family | ||||||||
| Protein Function |
This alpha-adrenergic receptor mediates its action by association with G proteins that activate a phosphatidylinositol-calcium second messenger system. Its effect is mediated by G(q) and G(11) proteins. Nuclear ADRA1A-ADRA1B heterooligomers regulate phenylephrine(PE)-stimulated ERK signaling in cardiac myocytes.
Click to Show/Hide
|
||||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Prostaglandin G/H synthase 2 |
×
Structure
Sequence
MLARALLLCAVLALSHTANPCCSHPCQNRGVCMSVGFDQYKCDCTRTGFYGENCSTPEFLTRIKLFLKPTPNTVHYILTHFKGFWNVVNNIPFLRNAIMSYVLTSRSHLIDSPPTYNADYGYKSWEAFSNLSYYTRALPPVPDDCPTPLGVKGKKQLPDSNEIVEKLLLRRKFIPDPQGSNMMFAFFAQHFTHQFFKTDHKRGPAFTNGLGHGVDLNHIYGETLARQRKLRLFKDGKMKYQIIDGEMYPPTVKDTQAEMIYPPQVPEHLRFAVGQEVFGLVPGLMMYATIWLREHNRVCDVLKQEHPEWGDEQLFQTSRLILIGETIKIVIEDYVQHLSGYHFKLKFDPELLFNKQFQYQNRIAAEFNTLYHWHPLLPDTFQIHDQKYNYQQFIYNNSILLEHGITQFVESFTRQIAGRVAGGRNVPPAVQKVSQASIDQSRQMKYQSFNEYRKRFMLKPYESFEELTGEKEMSAELEALYGDIDAVELYPALLVEKPRPDAIFGETMVEVGAPFSLKGLMGNVICSPAYWKPSTFGGEVGFQIINTASIQSLICNNVKGCPFTSFSVPDPELIKTVTINASSSRSGLDDINPTVLLKERSTEL
|
|||||||
| Gene Name | PTGS2 | ||||||||
| Uniprot ID | PGH2_HUMAN | ||||||||
| KEGG Pathway | hsa:5743 | ||||||||
| Protein Family | Prostaglandin G/H synthase family | ||||||||
| Protein Function |
Dual cyclooxygenase and peroxidase in the biosynthesis pathway of prostanoids, a class of C20 oxylipins mainly derived from arachidonate, with a particular role in the inflammatory response (PubMed:7947975, PubMed:7592599, PubMed:9261177, PubMed:16373578, PubMed:22942274, PubMed:26859324, PubMed:27226593, PubMed:11939906, PubMed:19540099). The cyclooxygenase activity oxygenates arachidonate (AA, C20:4(n-6)) to the hydroperoxy endoperoxide prostaglandin G2 (PGG2), and the peroxidase activity reduces PGG2 to the hydroxy endoperoxide PGH2, the precursor of all 2-series prostaglandins and thromboxanes (PubMed:7947975, PubMed:7592599, PubMed:9261177, PubMed:16373578, PubMed:22942274, PubMed:26859324, PubMed:27226593). This complex transformation is initiated by abstraction of hydrogen at carbon 13 (with S-stereochemistry), followed by insertion of molecular O2 to form the endoperoxide bridge between carbon 9 and 11 that defines prostaglandins. The insertion of a second molecule of O2 (bis-oxygenase activity) yields a hydroperoxy group in PGG2 that is then reduced to PGH2 by two electrons (PubMed:7947975, PubMed:7592599, PubMed:9261177, PubMed:16373578, PubMed:22942274, PubMed:26859324, PubMed:27226593). Similarly catalyzes successive cyclooxygenation and peroxidation of dihomo-gamma-linoleate (DGLA, C20:3(n-6)) and eicosapentaenoate (EPA, C20:5(n-3)) to corresponding PGH1 and PGH3, the precursors of 1- and 3-series prostaglandins (PubMed:11939906, PubMed:19540099). In an alternative pathway of prostanoid biosynthesis, converts 2-arachidonoyl lysophopholipids to prostanoid lysophopholipids, which are then hydrolyzed by intracellular phospholipases to release free prostanoids (PubMed:27642067). Metabolizes 2-arachidonoyl glycerol yielding the glyceryl ester of PGH2, a process that can contribute to pain response (PubMed:22942274). Generates lipid mediators from n-3 and n-6 polyunsaturated fatty acids (PUFAs) via a lipoxygenase-type mechanism. Oxygenates PUFAs to hydroperoxy compounds and then reduces them to corresponding alcohols (PubMed:11034610, PubMed:11192938, PubMed:9048568, PubMed:9261177). Plays a role in the generation of resolution phase interaction products (resolvins) during both sterile and infectious inflammation (PubMed:12391014). Metabolizes docosahexaenoate (DHA, C22:6(n-3)) to 17R-HDHA, a precursor of the D-series resolvins (RvDs) (PubMed:12391014). As a component of the biosynthetic pathway of E-series resolvins (RvEs), converts eicosapentaenoate (EPA, C20:5(n-3)) primarily to 18S-HEPE that is further metabolized by ALOX5 and LTA4H to generate 18S-RvE1 and 18S-RvE2 (PubMed:21206090). In vascular endothelial cells, converts docosapentaenoate (DPA, C22:5(n-3)) to 13R-HDPA, a precursor for 13-series resolvins (RvTs) shown to activate macrophage phagocytosis during bacterial infection (PubMed:26236990). In activated leukocytes, contributes to oxygenation of hydroxyeicosatetraenoates (HETE) to diHETES (5,15-diHETE and 5,11-diHETE) (PubMed:22068350, PubMed:26282205). During neuroinflammation, plays a role in neuronal secretion of specialized preresolving mediators (SPMs) 15R-lipoxin A4 that regulates phagocytic microglia (By similarity).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The patient's blood pressure should be monitored during coadministration, and the dosage adjusted as necessary. | ||||||||
