Details of Drug-Drug Interaction
| Drug General Information (ID: DDIFUI7BDZ) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Anti-inhibitor coagulant complex | Drug Info | Emicizumab | Drug Info | |||||
| Drug Type | Protein/peptide | Monoclonal antibody | |||||||
| Therapeutic Class | Coagulation Modifiers | Coagulation Modifiers | |||||||
| Mechanism of Anti-inhibitor coagulant complex-Emicizumab Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
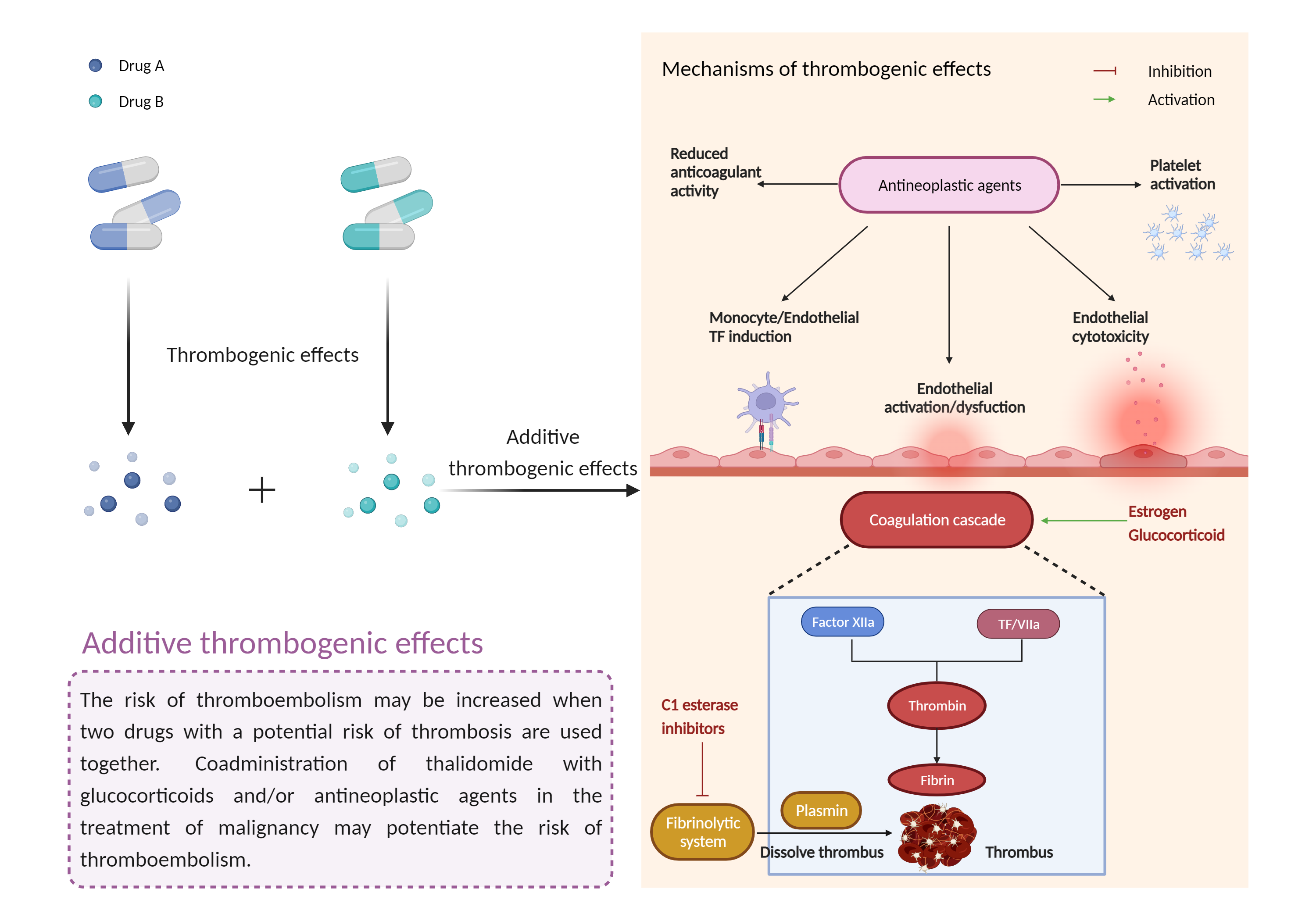
| Additive thrombogenic effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Anti-inhibitor coagulant complex | Emicizumab | |||||||
| Mechanism | Thrombogenic effects | Thrombogenic effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Thrombogenic effects | ||||||||
| Factor Description | Drug-induced thrombosis disrupts the balance between thrombogenic factors and protective mechanisms and tends to produce thrombosis. Drug-induced thrombosis usually causes venous thrombotic events, and arterial thrombotic events are also noted as a result of drug administration | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Close monitoring is recommended when activated prothrombin complex concentrate must be used during emicizumab prophylaxis. Immediately discontinue aPCC and suspend dosing of emicizumab if clinical symptoms, imaging, and/or laboratory findings consistent with TMA or thromboembolism occur, and manage as clinically indicated. Consider the benefits and risks of resuming emicizumab prophylaxis following complete resolution of TMA or thromboembolism on a case-by-case basis. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Hemlibra (emicizumab). Genentech, South San Francisco, CA. | ||||||||||||||||||
