Details of Drug-Drug Interaction
| Drug General Information (ID: DDIEZ3H8KP) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Aminoglutethimide | Drug Info | Ramelteon | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antineoplastics | Anxiolytics/Sedatives/Hypnotics | |||||||
| Structure | |||||||||
| Mechanism of Aminoglutethimide-Ramelteon Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| CYP450 enzyme induction Click to Show/Hide Mechanism Graph | |||||||||
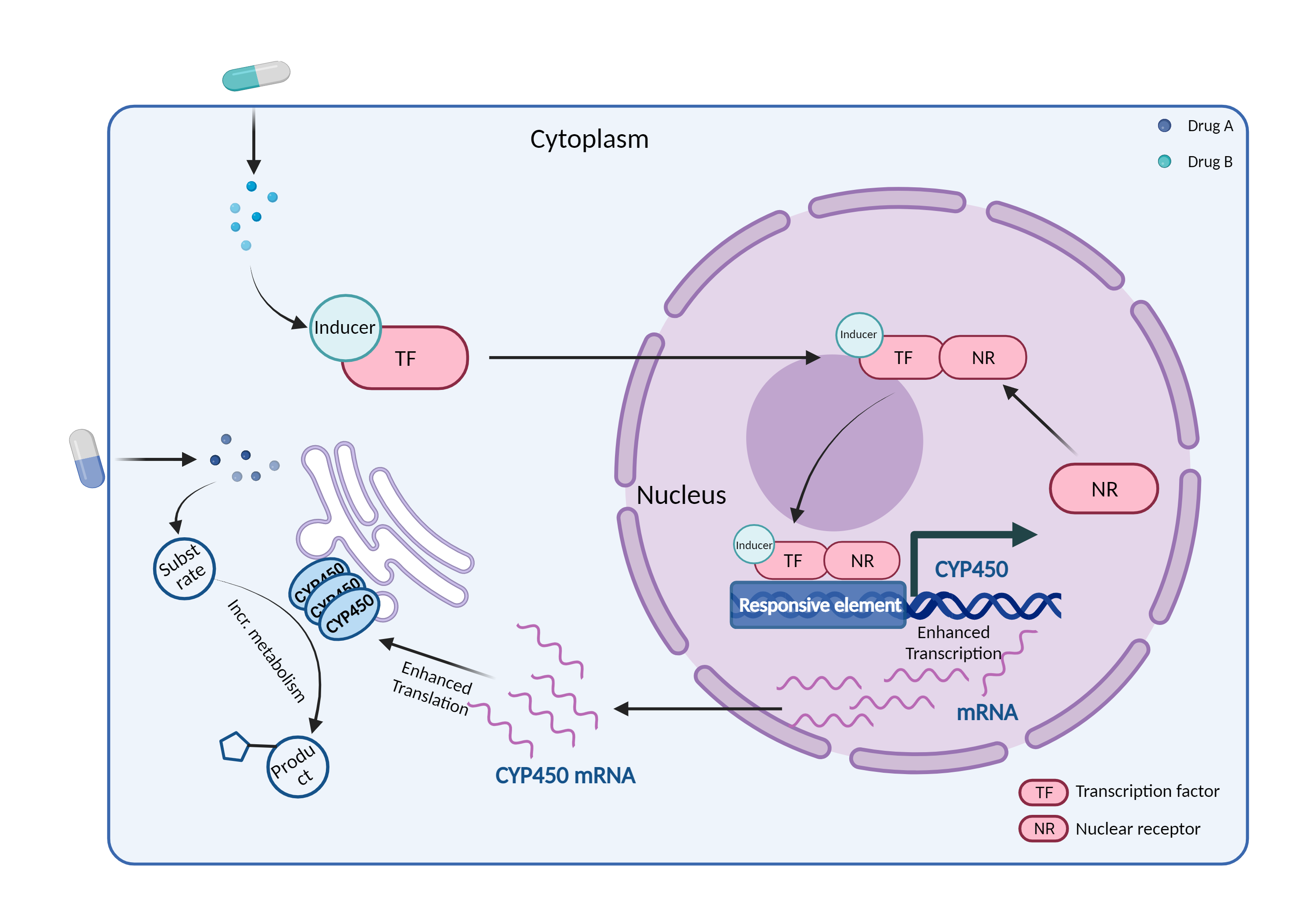 |
|||||||||
| Drug Name | Aminoglutethimide | Ramelteon | |||||||
| Mechanism 1 | CYP450 2C19 inducer | CYP450 2C19 substrate | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Cytochrome P450 2C19 |
×
Structure
Sequence
MDPFVVLVLCLSCLLLLSIWRQSSGRGKLPPGPTPLPVIGNILQIDIKDVSKSLTNLSKIYGPVFTLYFGLERMVVLHGYEVVKEALIDLGEEFSGRGHFPLAERANRGFGIVFSNGKRWKEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICSIIFQKRFDYKDQQFLNLMEKLNENIRIVSTPWIQICNNFPTIIDYFPGTHNKLLKNLAFMESDILEKVKEHQESMDINNPRDFIDCFLIKMEKEKQNQQSEFTIENLVITAADLLGAGTETTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRGHMPYTDAVVHEVQRYIDLIPTSLPHAVTCDVKFRNYLIPKGTTILTSLTSVLHDNKEFPNPEMFDPRHFLDEGGNFKKSNYFMPFSAGKRICVGEGLARMELFLFLTFILQNFNLKSLIDPKDLDTTPVVNGFASVPPFYQLCFIPV
|
|||||||
| Gene Name | CYP2C19 | ||||||||
| Uniprot ID | CP2CJ_HUMAN | ||||||||
| KEGG Pathway | hsa:1557 | ||||||||
| Protein Family | Cytochrome P450 family | ||||||||
| Protein Function |
A cytochrome P450 monooxygenase involved in the metabolism of polyunsaturated fatty acids (PUFA) (PubMed:18577768, PubMed:19965576, PubMed:20972997). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase) (PubMed:18577768, PubMed:19965576, PubMed:20972997). Catalyzes the hydroxylation of carbon-hydrogen bonds. Hydroxylates PUFA specifically at the omega-1 position (PubMed:18577768). Catalyzes the epoxidation of double bonds of PUFA (PubMed:20972997, PubMed:19965576). Also metabolizes plant monoterpenes such as limonene. Oxygenates (R)- and (S)-limonene to produce carveol and perillyl alcohol (PubMed:11950794). Responsible for the metabolism of a number of therapeutic agents such as the anticonvulsant drug S-mephenytoin, omeprazole, proguanil, certain barbiturates, diazepam, propranolol, citalopram and imipramine. Hydroxylates fenbendazole at the 4' position (PubMed:23959307).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Mechanism 2 | CYP450 3A4 inducer | CYP450 3A4 substrate | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Cytochrome P450 3A4 |
×
Structure
Sequence
MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMFDMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISIAEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYSMDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICVFPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSIIFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVVNETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFSKKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLGGLLQPEKPVVLKVESRDGTVSGA
|
|||||||
| Gene Name | CYP3A4 | ||||||||
| Uniprot ID | CP3A4_HUMAN | ||||||||
| KEGG Pathway | hsa:1576 | ||||||||
| Protein Family | Cytochrome P450 family | ||||||||
| Protein Function |
A cytochrome P450 monooxygenase involved in the metabolism of sterols, steroid hormones, retinoids and fatty acids (PubMed:10681376, PubMed:11093772, PubMed:11555828, PubMed:14559847, PubMed:12865317, PubMed:15373842, PubMed:15764715, PubMed:20702771, PubMed:19965576, PubMed:21490593, PubMed:21576599). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase). Catalyzes the hydroxylation of carbon-hydrogen bonds (PubMed:2732228, PubMed:14559847, PubMed:12865317, PubMed:15373842, PubMed:15764715, PubMed:21576599, PubMed:21490593). Exhibits high catalytic activity for the formation of hydroxyestrogens from estrone (E1) and 17beta-estradiol (E2), namely 2-hydroxy E1 and E2, as well as D-ring hydroxylated E1 and E2 at the C-16 position (PubMed:11555828, PubMed:14559847, PubMed:12865317). Plays a role in the metabolism of androgens, particularly in oxidative deactivation of testosterone (PubMed:2732228, PubMed:15373842, PubMed:15764715, PubMed:22773874). Metabolizes testosterone to less biologically active 2beta- and 6beta-hydroxytestosterones (PubMed:2732228, PubMed:15373842, PubMed:15764715). Contributes to the formation of hydroxycholesterols (oxysterols), particularly A-ring hydroxylated cholesterol at the C-4beta position, and side chain hydroxylated cholesterol at the C-25 position, likely contributing to cholesterol degradation and bile acid biosynthesis (PubMed:21576599). Catalyzes bisallylic hydroxylation of polyunsaturated fatty acids (PUFA) (PubMed:9435160). Catalyzes the epoxidation of double bonds of PUFA with a preference for the last double bond (PubMed:19965576). Metabolizes endocannabinoid arachidonoylethanolamide (anandamide) to 8,9-, 11,12-, and 14,15-epoxyeicosatrienoic acid ethanolamides (EpETrE-EAs), potentially modulating endocannabinoid system signaling (PubMed:20702771). Plays a role in the metabolism of retinoids. Displays high catalytic activity for oxidation of all-trans-retinol to all-trans-retinal, a rate-limiting step for the biosynthesis of all-trans-retinoic acid (atRA) (PubMed:10681376). Further metabolizes atRA toward 4-hydroxyretinoate and may play a role in hepatic atRA clearance (PubMed:11093772). Responsible for oxidative metabolism of xenobiotics. Acts as a 2-exo-monooxygenase for plant lipid 1,8-cineole (eucalyptol) (PubMed:11159812). Metabolizes the majority of the administered drugs. Catalyzes sulfoxidation of the anthelmintics albendazole and fenbendazole (PubMed:10759686). Hydroxylates antimalarial drug quinine (PubMed:8968357). Acts as a 1,4-cineole 2-exo-monooxygenase (PubMed:11695850). Also involved in vitamin D catabolism and calcium homeostasis. Catalyzes the inactivation of the active hormone calcitriol (1-alpha,25-dihydroxyvitamin D(3)) (PubMed:29461981).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The efficacy of ramelteon may be reduced when prescribed with potent inducers of CYP450 isoenzymes such as carbamazepine, enzalutamide, phenobarbital, phenytoin, rifampin, and St. John's wort. Other known inducers include aminoglutethimide, bexarotene, bosentan, dabrafenib, dexamethasone, efavirenz, etravirine, mitotane, modafinil, nafcillin, nevirapine, rifabutin, rifapentine, barbiturates and various other anticonvulsants, although the extent to which they interact with ramelteon is unknown. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Rozerem (ramelteon). Takeda Pharmaceuticals America, Lincolnshire, IL. | ||||||||||||||||||
