Details of Drug-Drug Interaction
| Drug General Information (ID: DDIE5U6GI4) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Nabumetone | Drug Info | Clofarabine | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Nsaids/Analgesics | Antineoplastics | |||||||
| Structure | |||||||||
| Mechanism of Nabumetone-Clofarabine Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
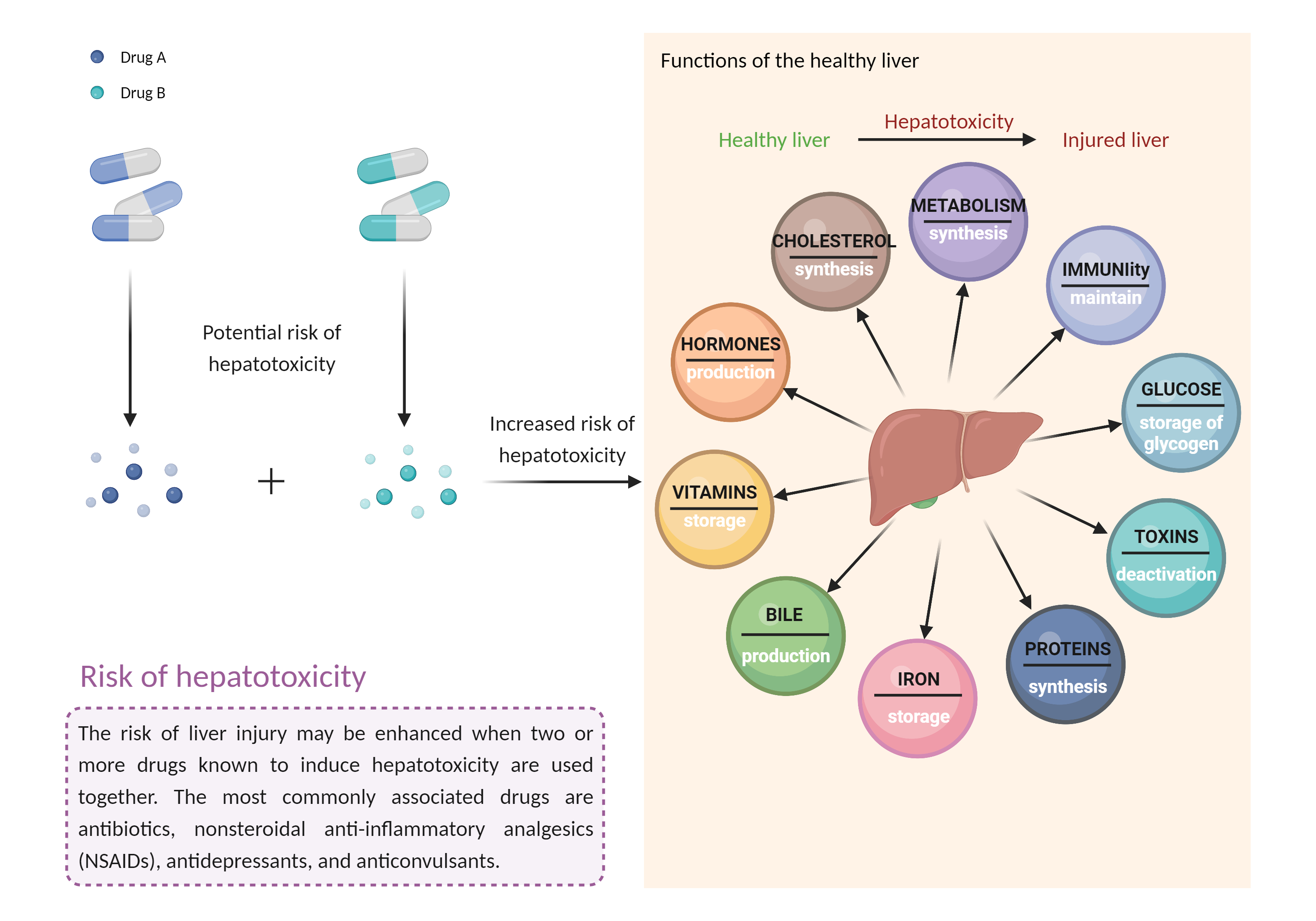
| Increased risk of hepatotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Nabumetone | Clofarabine | |||||||
| Mechanism 1 | Hepatotoxicity | Hepatotoxicity | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hepatotoxicity | ||||||||
| Factor Description | Combination of drugs that can induce hepatotoxicity may increase the risk of liver injury. Symptoms vary depending on the level of exposure and the total extent of liver damage, and may cause few symptoms if the damage is mild, and eventually lead to liver failure in patients with severe damage. | ||||||||
| Mechanism Description |
|
||||||||
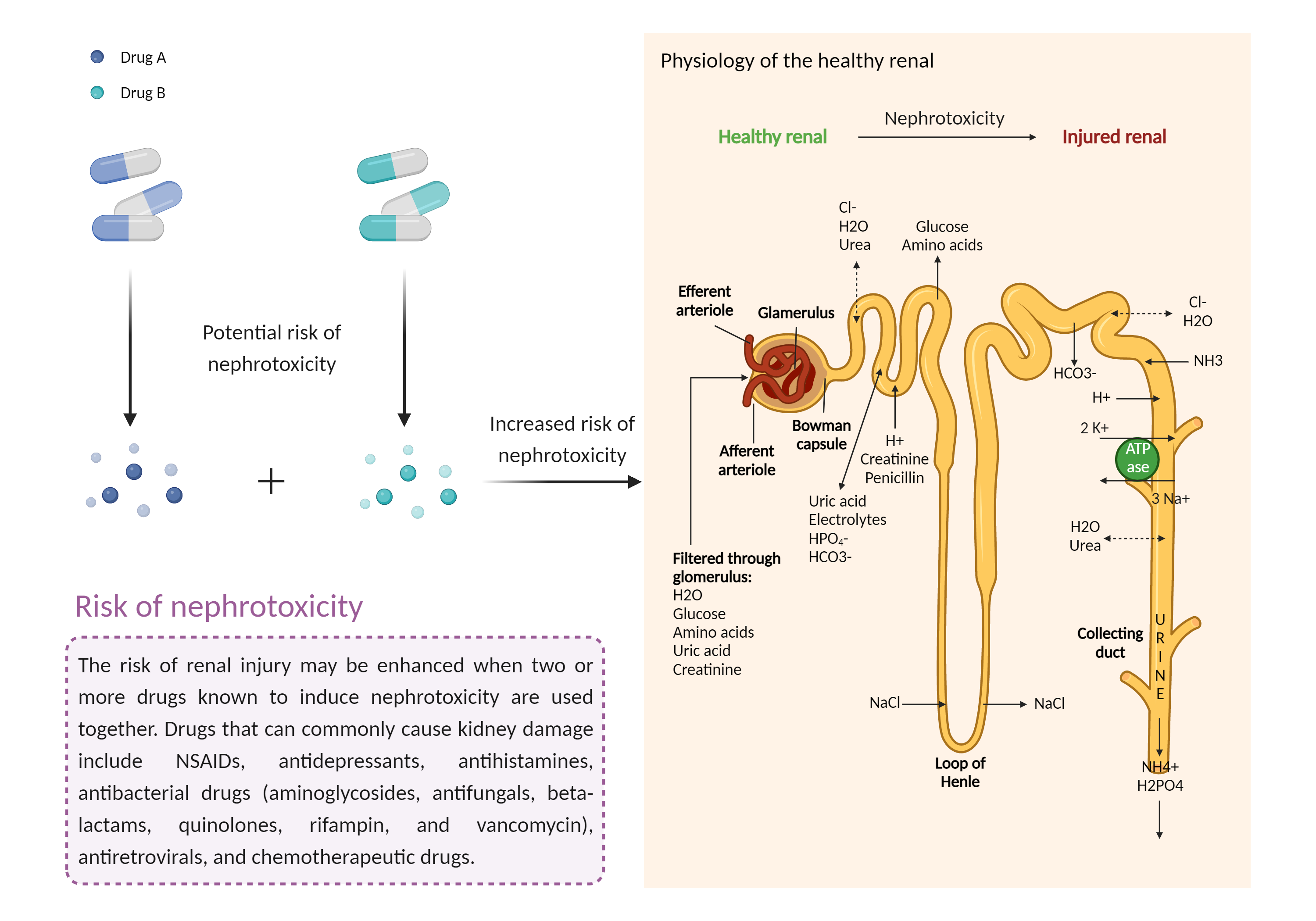
| Increased risk of nephrotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Nabumetone | Clofarabine | |||||||
| Mechanism 2 | Nephrotoxicity | Nephrotoxicity | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Nephrotoxicity | ||||||||
| Factor Description | The combination of drugs that can induce nephrotoxicity may increase the risk of kidney injury. When kidney injury occurs, the inability to remove excess urine and waste from the body can lead to high levels of urea nitrogen, creatinine, and electrolytes (such as potassium and magnesium) in the blood. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | If possible, drugs that are potentially nephrotoxic including NSAIDs should be avoided during the 5 days of clofarabine administration. Because NSAIDs can also be hepatotoxic, it may be appropriate to avoid them altogether in some patients during clofarabine therapy. If an NSAID is required, ibuprofen appears to have the highest liver safety profile among NSAIDs, while diclofenac and sulindac have been linked to a higher frequency of liver damage. Patients treated with clofarabine should be advised to seek medical attention if they experience potential signs and symptoms of hepatotoxicity such as fever, rash, itching, anorexia, nausea, vomiting, fatigue, malaise, right upper quadrant pain, dark urine, pale stools, and jaundice. Renal and hepatic function should be monitored prior to and daily during clofarabine therapy, and administration discontinued immediately if substantial increases (e.g., grade 3 or higher) in creatinine, liver enzymes, or bilirubin are noted. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||||||||||||||||
| 2 | Product Information. Clolar (clofarabine). sanofi-aventis, Bridgewater, NJ. | ||||||||||||||||||
