| Mechanism of Elotuzumab-Teriflunomide Interaction
(Severity Level: Major)
|
|
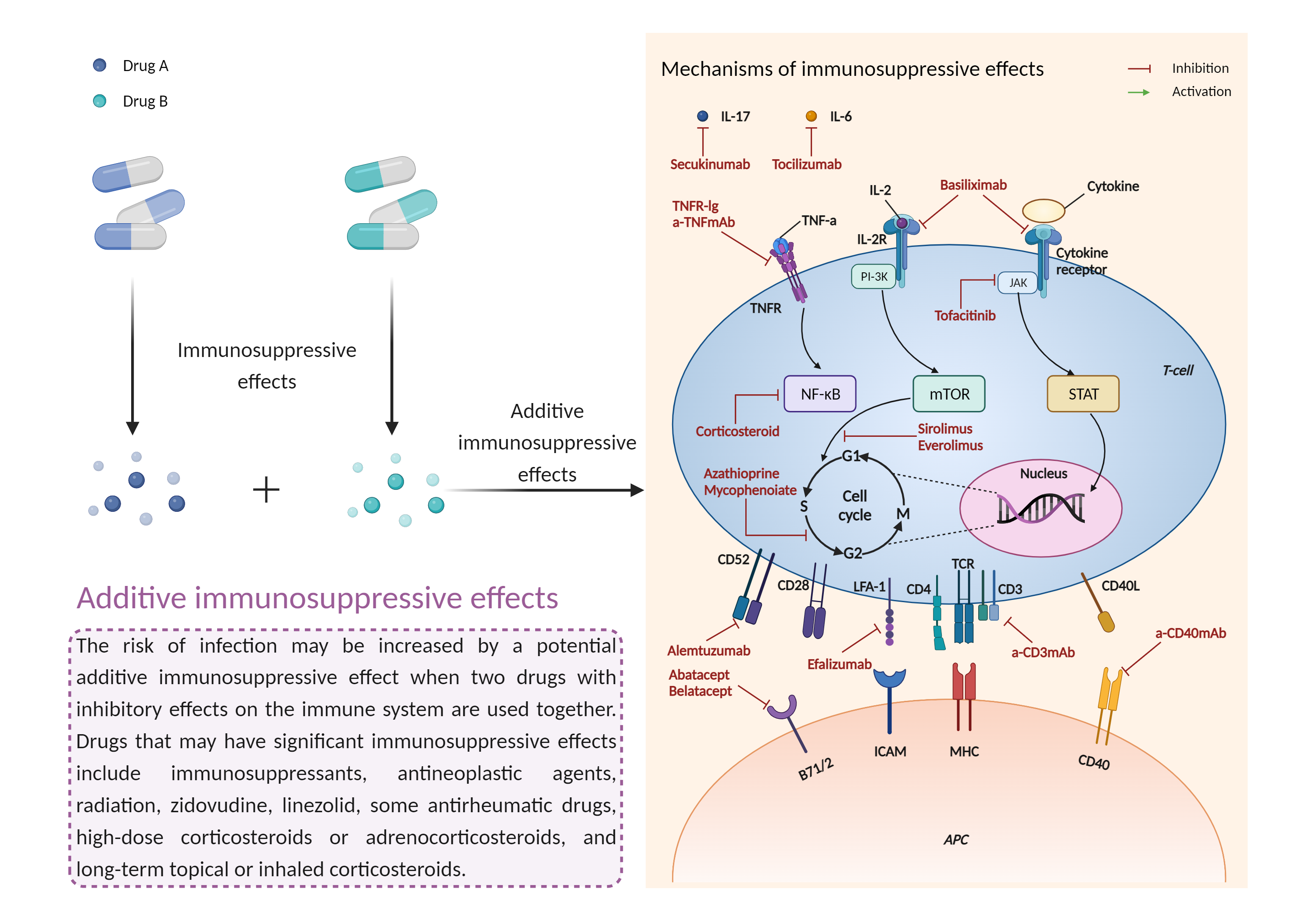
Additive immunosuppressive effects
Click to Show/Hide Mechanism Graph
|
 |
| Drug Name |
Elotuzumab |
Teriflunomide |
|
Mechanism 1
|
Immunosuppressive effects |
Immunosuppressive effects |
| Key Mechanism Factor 1 |
| Factor Name |
Immunosuppressive effects |
| Factor Description |
Immunosuppression is when your immune system is not functioning as it should. The immune system is made up of cells, tissues and organs that help the body fight off infections. If the immune system is suppressed, an infection that your body was able to control may become serious or even fatal. |
| Mechanism Description |
- Additive immunosuppressive effects by the combination of Elotuzumab and Teriflunomide
|
|
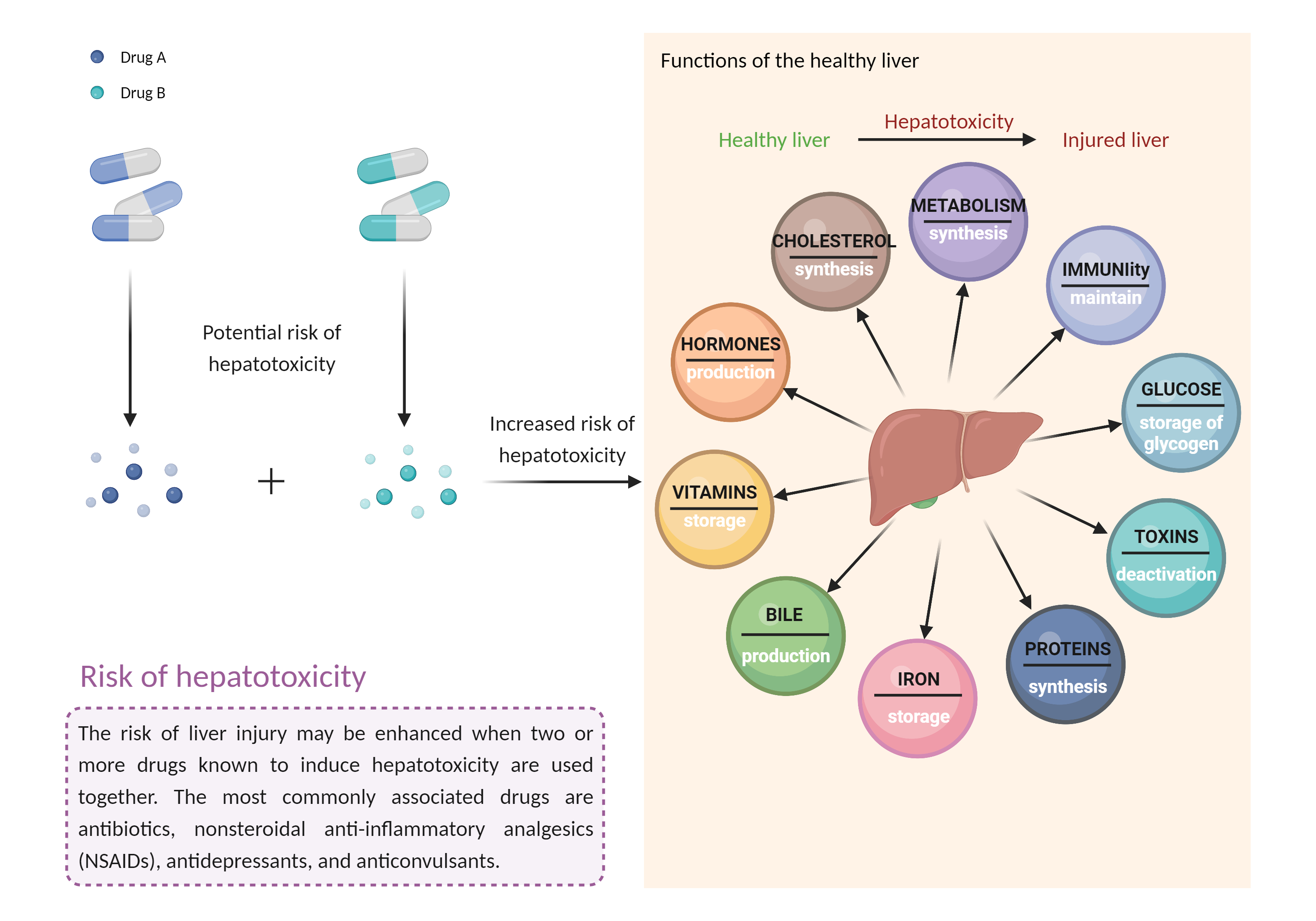
Increased risk of hepatotoxicity
Click to Show/Hide Mechanism Graph
|
 |
| Drug Name |
Elotuzumab |
Teriflunomide |
|
Mechanism 2
|
Hepatotoxicity |
Hepatotoxicity |
| Key Mechanism Factor 2 |
| Factor Name |
Hepatotoxicity |
| Factor Description |
Combination of drugs that can induce hepatotoxicity may increase the risk of liver injury. Symptoms vary depending on the level of exposure and the total extent of liver damage, and may cause few symptoms if the damage is mild, and eventually lead to liver failure in patients with severe damage. |
| Mechanism Description |
- Increased risk of hepatotoxicity by the combination of Elotuzumab and Teriflunomide
|


