Details of Drug-Drug Interaction
| Drug General Information (ID: DDICI9482L) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Cefdinir | Drug Info | Ferrous sulfate anhydrous | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antibiotics | Iron Supplement | |||||||
| Structure | |||||||||
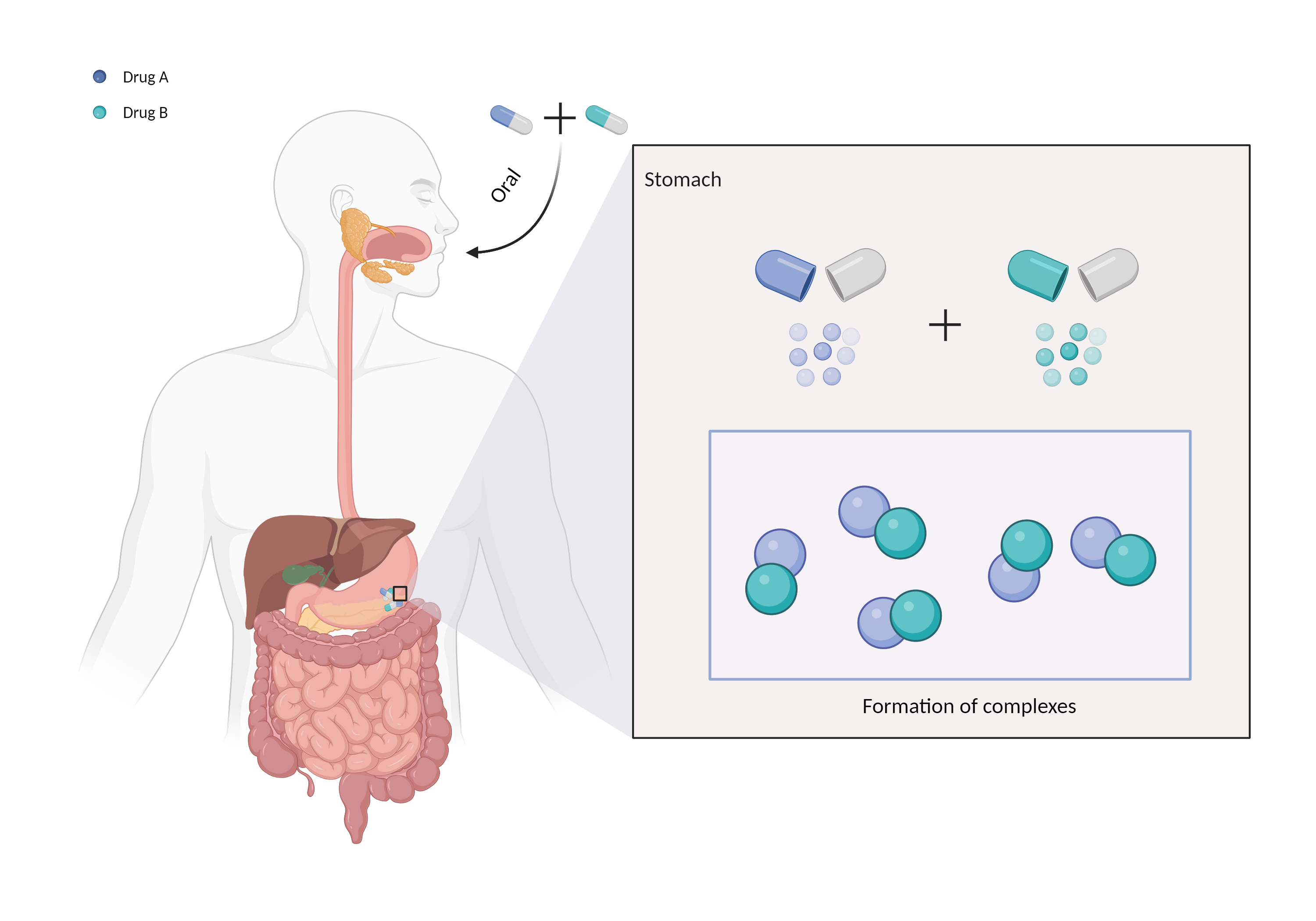
| Mechanism of Cefdinir-Ferrous sulfate anhydrous Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Complex formation Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Cefdinir | Ferrous sulfate anhydrous | |||||||
| Mechanism | Binds to polyvalent cations | Polyvalent cations | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Chelation | ||||||||
| Factor Description | Chelation is a direct drug interaction that usually involves the formation of dimers or trimers, resulting in larger complexes and poorer absorption. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | To minimize potential interaction with iron, the product labeling recommends that cefdinir be taken at least 2 hours before or 2 hours after administration of iron-containing products. However, this separation time may not be sufficient to prevent the interaction when sustained-release iron products are used, especially in higher dosages. Patients should be advised that reddish stools may occur due to formation of a nonabsorbable complex between cefdinir or its breakdown products and iron in the gastrointestinal tract. | ||||||||
