Details of Drug-Drug Interaction
| Drug General Information (ID: DDIBPJ3GDC) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Gefitinib | Drug Info | Dexlansoprazole | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antineoplastics | Proton Pump Inhibitors | |||||||
| Structure | |||||||||
| Mechanism of Gefitinib-Dexlansoprazole Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
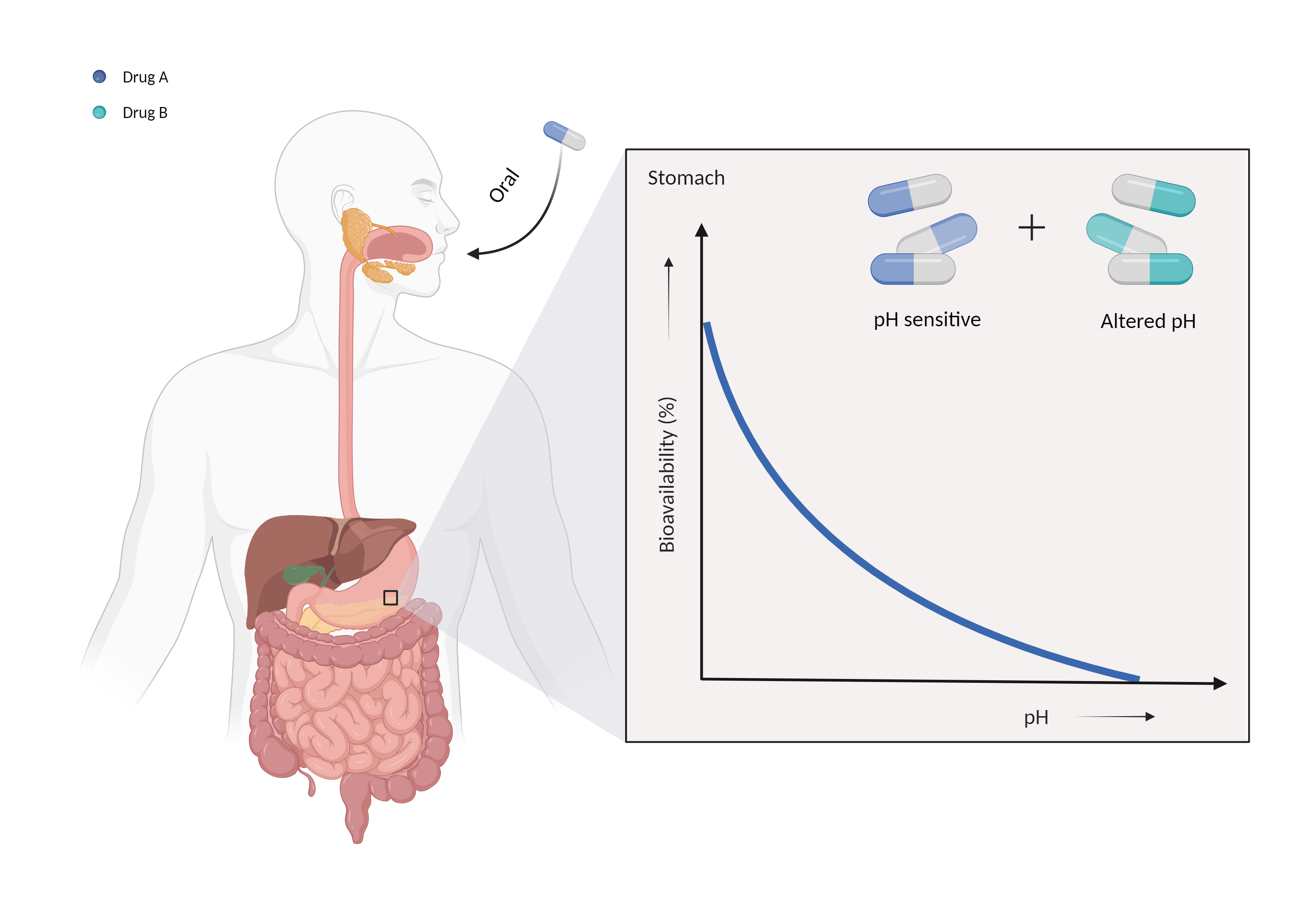
| Altered gastric pH Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Gefitinib | Dexlansoprazole | |||||||
| Mechanism | Gastric pH sensitive | Gastric alkalinizer | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Gastric pH | ||||||||
| Factor Description | The normal pH range of gastric acid is between 1.5 and 3.5 and is highly acidic, consisting mainly of hydrochloric acid. Changes in the pH of the stomach can alter the absorption of drugs. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The possibility of a diminished therapeutic response to gefitinib should be considered during coadministration with H2-receptor antagonists or proton pump inhibitors. Pharmacologic response to gefitinib should be monitored more closely whenever one of these agents is added to or withdrawn from therapy. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Iressa (gefitinib). Astra-Zeneca Pharmaceuticals, Wilmington, DE. | ||||||||||||||||||
