Details of Drug-Drug Interaction
| Drug General Information (ID: DDIBI38QN9) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Mechlorethamine | Drug Info | Thalidomide | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antineoplastics | Immunosuppressive Agents | |||||||
| Structure | |||||||||
| Mechanism of Mechlorethamine-Thalidomide Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
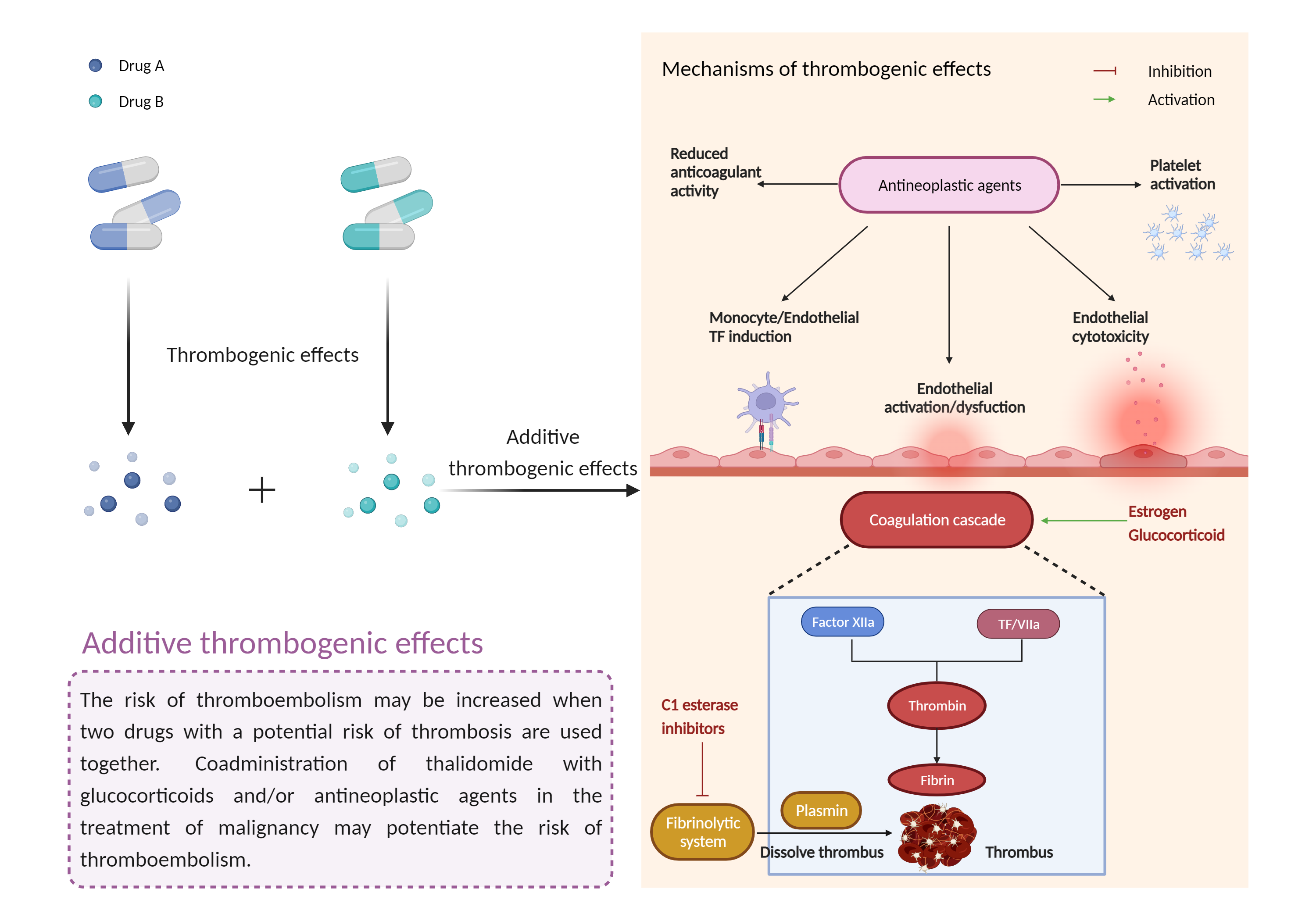
| Additive thrombogenic effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Mechlorethamine | Thalidomide | |||||||
| Mechanism | Thrombogenic effects | Thrombogenic effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Thrombogenic effects | ||||||||
| Factor Description | Drug-induced thrombosis disrupts the balance between thrombogenic factors and protective mechanisms and tends to produce thrombosis. Drug-induced thrombosis usually causes venous thrombotic events, and arterial thrombotic events are also noted as a result of drug administration | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Close monitoring for DVT or pulmonary embolism is recommended in patients who require thalidomide therapy in combination with glucocorticoids and/or cytotoxic agents. Patients should be advised to seek medical attention if they develop potential signs and symptoms of thromboembolism such as chest pain, shortness of breath, and pain or swelling in the arms or legs. Prophylaxis with anticoagulants such as low-molecular weight heparins or warfarin may be appropriate, but the decision to take thromboprophylactic measures should be made after careful assessment of underlying risk factors. If a thromboembolic event occurs during therapy with thalidomide, treatment must be discontinued and standard anticoagulation therapy started. Once anticoagulation is stabilized and complications of the thromboembolic event under control, thalidomide may be restarted at the original dose if benefit is deemed to outweigh the risks. | ||||||||
