Details of Drug-Drug Interaction
| Drug General Information (ID: DDIANLJ92F) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Diethylstilbestrol | Drug Info | Valproic acid | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antineoplastics | Anticonvulsants | |||||||
| Structure | |||||||||
| Mechanism of Diethylstilbestrol-Valproic acid Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| UGT induction Click to Show/Hide Mechanism Graph | |||||||||
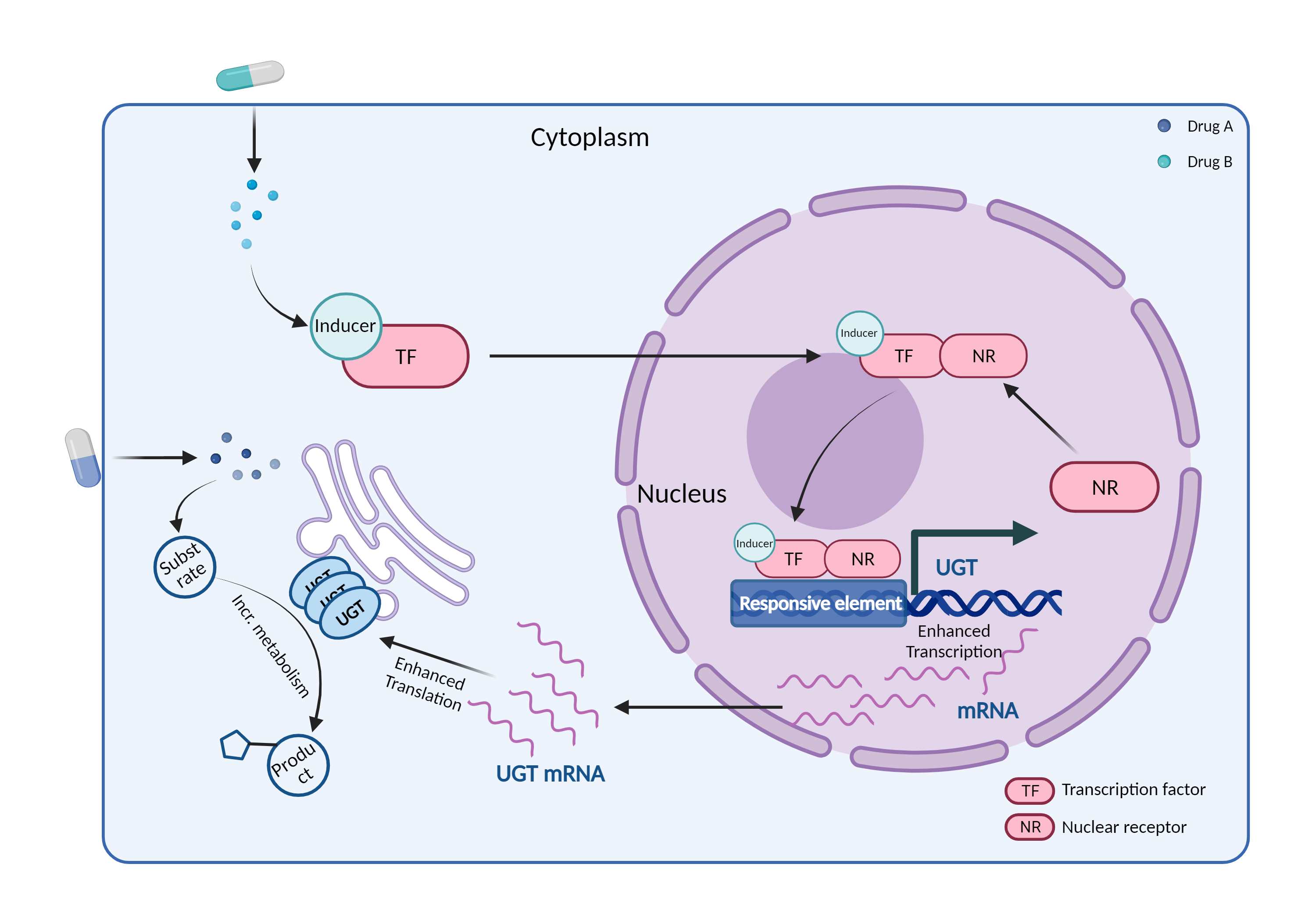 |
|||||||||
| Drug Name | Diethylstilbestrol | Valproic acid | |||||||
| Mechanism | UGT inducer | UGT substrate | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | UDP-glucuronosyltransferase | Structure Sequence | |||||||
| Protein Family | UDP-glycosyltransferase family | ||||||||
| Protein Function |
[Isoform 1]: UDP-glucuronosyltransferase (UGT) that catalyzes phase II biotransformation reactions in which lipophilic substrates are conjugated with glucuronic acid to increase the metabolite's water solubility, thereby facilitating excretion into either the urine or bile (PubMed:12181437, PubMed:15472229, PubMed:18004206, PubMed:18004212, PubMed:18719240, PubMed:19830808, PubMed:23288867). Essential for the elimination and detoxification of drugs, xenobiotics and endogenous compounds (PubMed:12181437, PubMed:18004206, PubMed:18004212). Catalyzes the glucuronidation of endogenous estrogen hormones such as estradiol, estrone and estriol (PubMed:15472229, PubMed:18719240, PubMed:23288867). Involved in the glucuronidation of bilirubin, a degradation product occurring in the normal catabolic pathway that breaks down heme in vertebrates (PubMed:17187418, PubMed:18004206, PubMed:19830808). Also catalyzes the glucuronidation the isoflavones genistein, daidzein, glycitein, formononetin, biochanin A and prunetin, which are phytoestrogens with anticancer and cardiovascular properties (PubMed:18052087, PubMed:19545173). Involved in the glucuronidation of the AGTR1 angiotensin receptor antagonist losartan, a drug which can inhibit the effect of angiotensin II (PubMed:18674515). Involved in the biotransformation of 7-ethyl-10-hydroxycamptothecin (SN-38), the pharmacologically active metabolite of the anticancer drug irinotecan (PubMed:12181437, PubMed:18004212, PubMed:20610558).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Pharmacologic response and serum valproic levels should be monitored more closely whenever estrogen- and/or progestin-containing drugs are added to or withdrawn from therapy, and the valproic acid dosage adjusted as necessary. Patients should be advised to contact their physician if they experience loss of seizure control or symptoms of valproic acid toxicity such as tremors, ataxia, nystagmus, increased seizures, and changes in mental status. In patients receiving oral contraceptives, gradual transient increases in valproic acid levels will likely occur during the pill-free week for women not also taking an enzyme-inducing drug (e.g., carbamazepine, phenytoin, phenobarbital, primidone, rifampin). The increase in valproic acid levels will be greater if the dose of valproic acid is increased in the few days before or during the pill-free week. | ||||||||
