| Mechanism of Octreotide-Semaglutide Interaction
(Severity Level: Moderate)
|
|
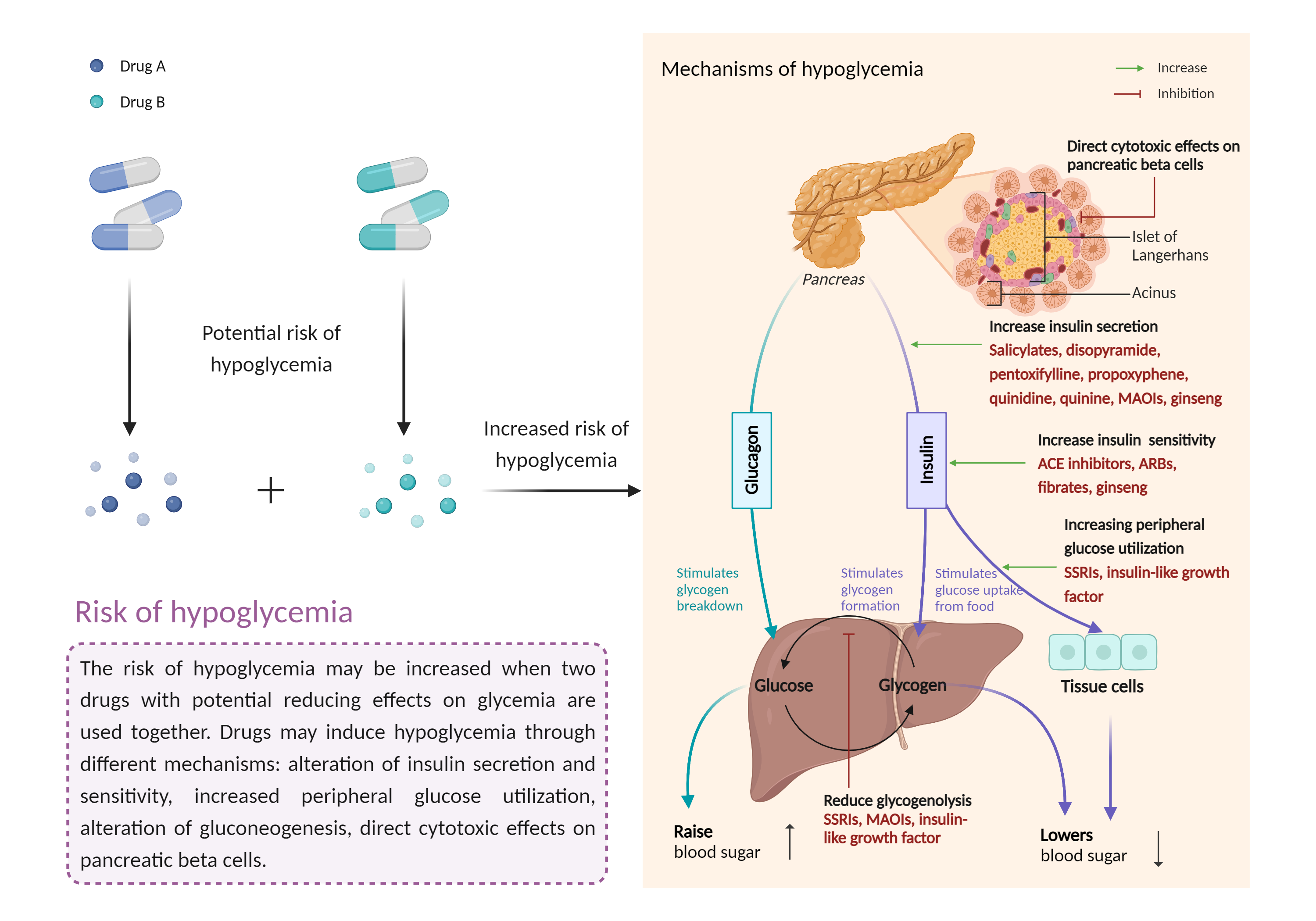
Increased risk of hypoglycemia
Click to Show/Hide Mechanism Graph
|
 |
| Drug Name |
Octreotide |
Semaglutide |
|
Mechanism 1
|
Hypoglycemic effects |
Antidiabetic agent |
| Key Mechanism Factor 1 |
| Factor Name |
Hypoglycemia |
| Factor Description |
Hypoglycemia is a condition in which your blood sugar (glucose) level is below the standard range. It causes irregular or rapid heartbeat, pale skin, numbness of the lips, tongue or cheeks, and sweating. |
| Mechanism Description |
- Increased risk of hypoglycemia by the combination of Octreotide and Semaglutide
|
|
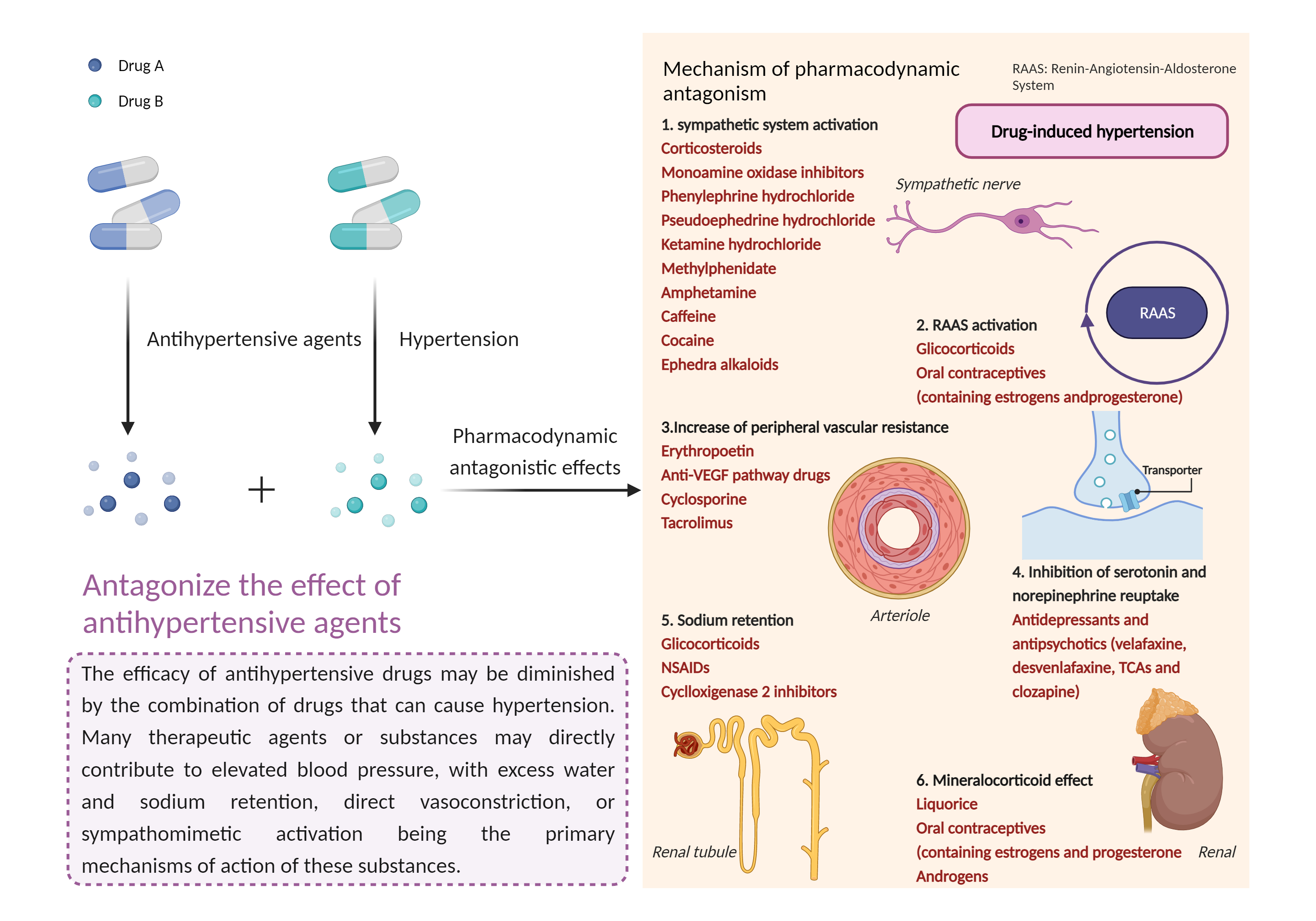
Antagonize the effect of antihypertensive agents
Click to Show/Hide Mechanism Graph
|
 |
| Drug Name |
Octreotide |
Semaglutide |
| Mechanism 2 |
Hyperglycemia |
Antidiabetic agent |
| Key Mechanism Factor 2 |
| Factor Name |
Antihypertensive agents |
| Factor Description |
The hypotensive effect of antihypertensive agents may be reduced, leading to higher blood pressure, increased blood pressure fluctuations, and possible cardiovascular damage. |
| Mechanism Description |
- Antagonize the effect of Semaglutide when combined with Octreotide
|
| Mechanism 3 |
Hyperglycemic effects |
Antidiabetic agent |
| Key Mechanism Factor 3 |
| Factor Name |
Antihypertensive agents |
| Factor Description |
The hypotensive effect of antihypertensive agents may be reduced, leading to higher blood pressure, increased blood pressure fluctuations, and possible cardiovascular damage. |
| Mechanism Description |
- Antagonize the effect of Semaglutide when combined with Octreotide
|


