Details of Drug-Drug Interaction
| Drug General Information (ID: DDI91YL8G7) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Sumatriptan | Drug Info | Sibutramine | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Vasoconstrictor Agents | Appetite Depressants | |||||||
| Structure | |||||||||
| Mechanism of Sumatriptan-Sibutramine Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Additive serotonergic effects Click to Show/Hide Mechanism Graph | |||||||||
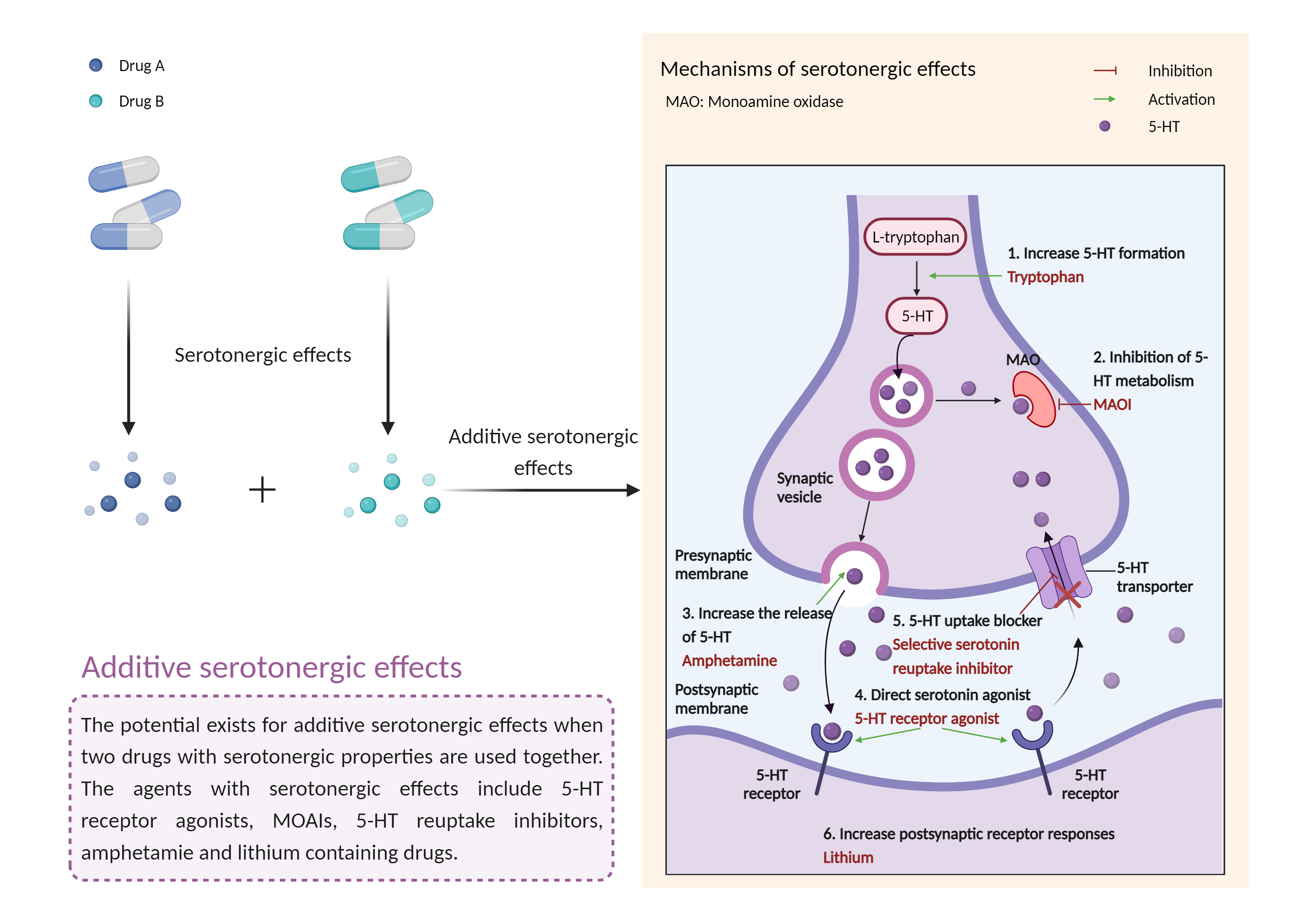 |
|||||||||
| Drug Name | Sumatriptan | Sibutramine | |||||||
| Mechanism |
Serotonergic effects 5-HT 1 receptor Agonist |
Serotonergic effects Serotonin transporter Inhibitor |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | 5-HT 1 receptor | Structure Sequence | |||||||
| Protein Family | G-protein coupled receptor 1 family | ||||||||
| Protein Function |
G-protein coupled receptor for 5-hydroxytryptamine (serotonin). Also functions as a receptor for various drugs and psychoactive substances. Ligand binding causes a conformation change that triggers signaling via guanine nucleotide-binding proteins (G proteins) and modulates the activity of down-stream effectors, such as adenylate cyclase. Beta-arrestin family members inhibit signaling via G proteins and mediate activation of alternative signaling pathways. Signaling inhibits adenylate cyclase activity and activates a phosphatidylinositol-calcium second messenger system that regulates the release of Ca(2+) ions from intracellular stores. Plays a role in the regulation of 5-hydroxytryptamine release and in the regulation of dopamine and 5-hydroxytryptamine metabolism. Plays a role in the regulation of dopamine and 5-hydroxytryptamine levels in the brain, and thereby affects neural activity, mood and behavior. Plays a role in the response to anxiogenic stimuli.
Click to Show/Hide
|
||||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Serotonin transporter |
×
Structure
Sequence
METTPLNSQKQLSACEDGEDCQENGVLQKVVPTPGDKVESGQISNGYSAVPSPGAGDDTRHSIPATTTTLVAELHQGERETWGKKVDFLLSVIGYAVDLGNVWRFPYICYQNGGGAFLLPYTIMAIFGGIPLFYMELALGQYHRNGCISIWRKICPIFKGIGYAICIIAFYIASYYNTIMAWALYYLISSFTDQLPWTSCKNSWNTGNCTNYFSEDNITWTLHSTSPAEEFYTRHVLQIHRSKGLQDLGGISWQLALCIMLIFTVIYFSIWKGVKTSGKVVWVTATFPYIILSVLLVRGATLPGAWRGVLFYLKPNWQKLLETGVWIDAAAQIFFSLGPGFGVLLAFASYNKFNNNCYQDALVTSVVNCMTSFVSGFVIFTVLGYMAEMRNEDVSEVAKDAGPSLLFITYAEAIANMPASTFFAIIFFLMLITLGLDSTFAGLEGVITAVLDEFPHVWAKRRERFVLAVVITCFFGSLVTLTFGGAYVVKLLEEYATGPAVLTVALIEAVAVSWFYGITQFCRDVKEMLGFSPGWFWRICWVAISPLFLLFIICSFLMSPPQLRLFQYNYPYWSIILGYCIGTSSFICIPTYIAYRLIITPGTFKERIIKSITPETPTEIPCGDIRLNAV
|
|||||||
| Gene Name | SLC6A4 | ||||||||
| Uniprot ID | SC6A4_HUMAN | ||||||||
| KEGG Pathway | hsa:6532 | ||||||||
| Protein Family | Sodium:neurotransmitter symporter (SNF) (TC 2.A.22) family | ||||||||
| Protein Function |
Serotonin transporter whose primary function in the central nervous system involves the regulation of serotonergic signaling via transport of serotonin molecules from the synaptic cleft back into the pre-synaptic terminal for re-utilization. Plays a key role in mediating regulation of the availability of serotonin to other receptors of serotonergic systems. Terminates the action of serotonin and recycles it in a sodium-dependent manner.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | In general, the concomitant use of sibutramine with other agents that affect the serotonergic neurotransmitter system should be avoided if possible, or otherwise approached with caution if potential benefit is deemed to outweigh the risk. Patients should be closely monitored for symptoms of the serotonin syndrome during treatment. Particular caution is advised when initiating or increasing the dosages of these agents. The potential risk for serotonin syndrome should be considered even when administering serotonergic agents sequentially, as some agents may demonstrate a prolonged elimination half-life. For example, a 5-week washout period is recommended following use of fluoxetine and 3 weeks following use of vortioxetine before administering another serotonergic agent such as sibutramine. At least 14 days should elapse between discontinuation of other serotonergic agents and initiation of treatment with sibutramine. If serotonin syndrome develops or is suspected during the course of therapy, all serotonergic agents should be discontinued immediately and supportive care rendered as necessary. Moderately ill patients may also benefit from the administration of a serotonin antagonist (e.g., cyproheptadine, chlorpromazine). Severe cases should be managed under consultation with a toxicologist and may require sedation, neuromuscular paralysis, intubation, and mechanical ventilation in addition to the other measures. | ||||||||
