Details of Drug-Drug Interaction
| Drug General Information (ID: DDI8ZA3JVS) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Pyridostigmine | Drug Info | Crizotinib | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Parasympathomimetics | Multikinase Inhibitors | |||||||
| Structure | |||||||||
| Mechanism of Pyridostigmine-Crizotinib Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
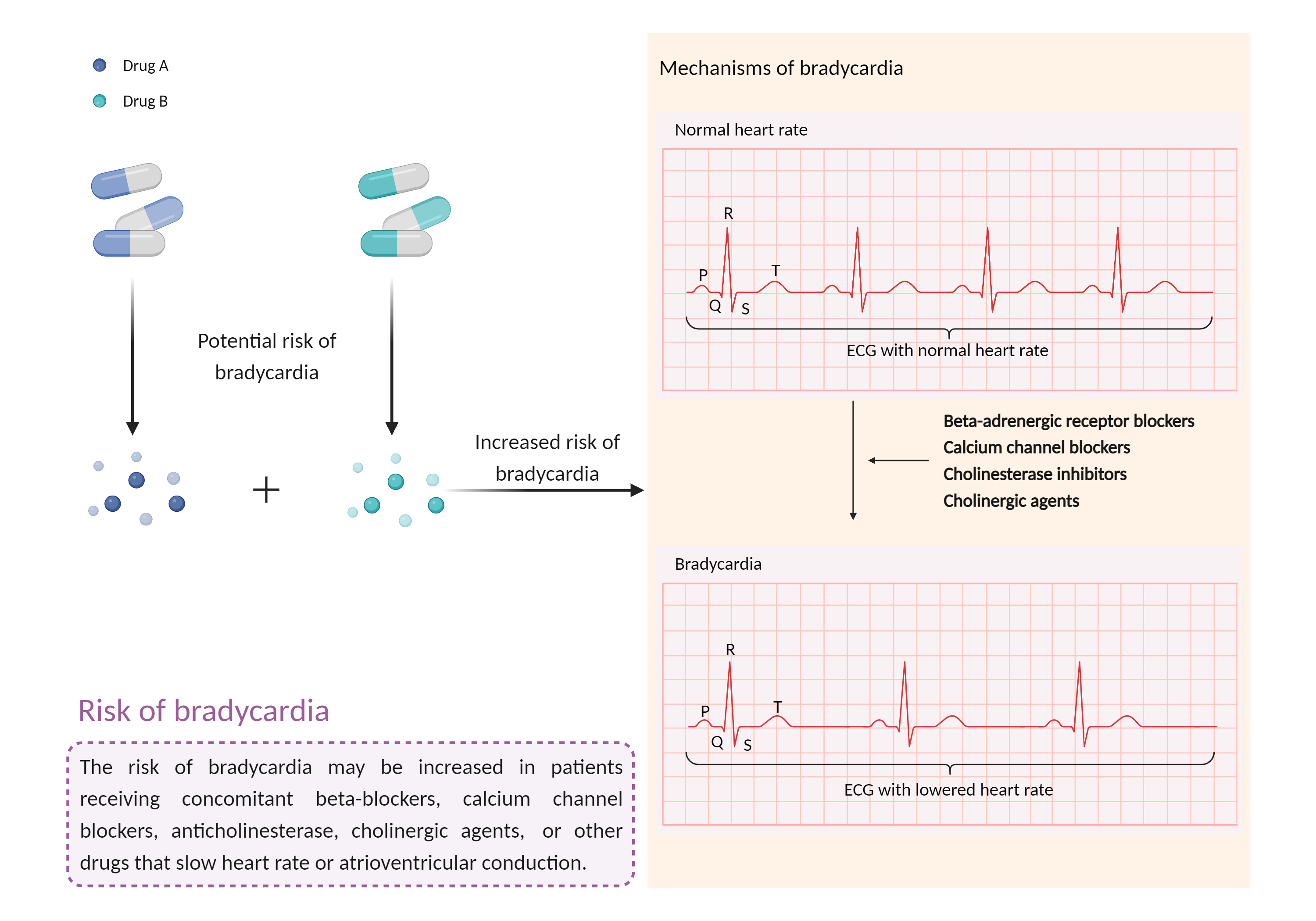
| Increased risk of bradycardia Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Pyridostigmine | Crizotinib | |||||||
| Mechanism | Bradycardia | Bradycardia | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Bradycardia | ||||||||
| Factor Description | Bradycardia is a slow heart rate in which the heart beats less than 60 times per minute. If the heart rate is very slow and the heart is not pumping enough oxygen-rich blood to the body, and you may feel dizzy, very tired or weak, and short of breath. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Concomitant use of crizotinib with other agents known to cause bradycardia such as beta-blockers, calcium channel blockers, cholinesterase inhibitors, clonidine, and digoxin should generally be avoided whenever possible. Otherwise, heart rate and blood pressure should be monitored closely. If Grade 2 or 3 symptomatic bradycardia occurs, crizotinib should be withheld, the use of concomitant medications re-evaluated, and the dosage of crizotinib adjusted accordingly. Crizotinib should be permanently discontinued for Grade 4 bradycardia. In patients who develop Grade 4 bradycardia associated with concomitant medications known to cause bradycardia or hypotension, crizotinib should be withheld until Grade 1 or less, and if concomitant medications can be discontinued, crizotinib may be restarted at 250 mg once daily with frequent monitoring. | ||||||||
