| Management |
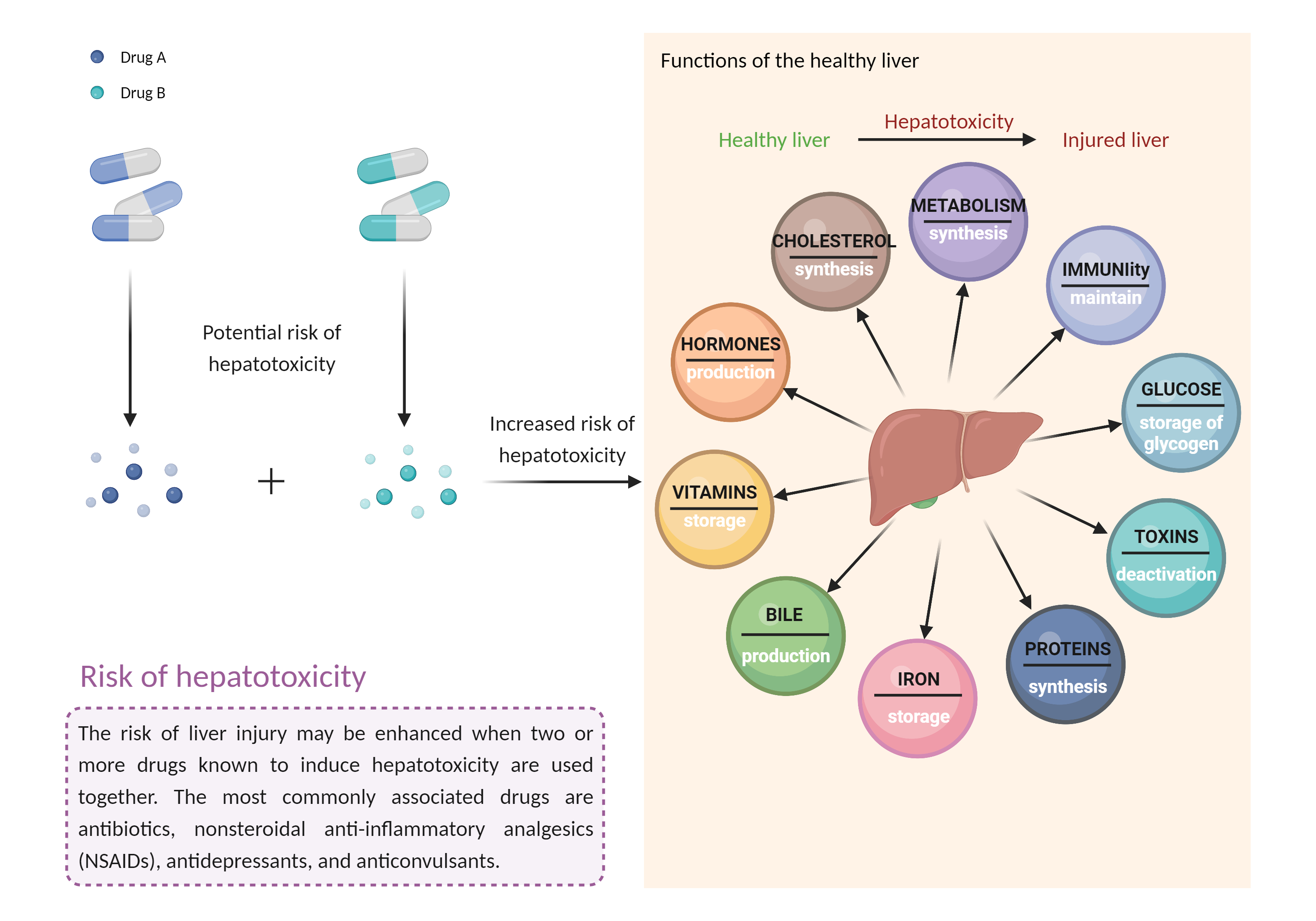
Caution is advised if lomitapide is used with other potentially hepatotoxic agents (e.g., acetaminophen alcohol amiodarone androgens and anabolic steroids antituberculous agents azole antifungal agents ACE inhibitors cyclosporine (high dosages) disulfiram endothelin receptor antagonists interferons ketolide and macrolide antibiotics kinase inhibitors methotrexate nonsteroidal anti-inflammatory agents nucleoside reverse transcriptase inhibitors proteasome inhibitors retinoids tamoxifen tetracyclines thiazolidinediones tolvaptan vincristine zileuton anticonvulsants such as carbamazepine, hydantoins, felbamate, and valproic acid other lipid-lowering medications such as fenofibrate, mipomersen, niacin, and statins herbals and nutritional supplements such as black cohosh, chaparral, comfrey, DHEA, kava, pennyroyal oil, and red yeast rice). Patients treated with lomitapide should have serum ALT, AST, alkaline phosphatase, and total bilirubin measured prior to initiation of treatment and regularly during treatment in accordance with the product labeling, and the dosing adjusted or interrupted as necessary. Since alcohol may increase levels of hepatic fat and induce or exacerbate liver injury, the manufacturer recommends that patients taking lomitapide not consume more than one alcoholic drink per day. Patients should be advised to seek medical attention if they experience potential signs and symptoms of hepatotoxicity such as fever, rash, itching, anorexia, nausea, vomiting, fatigue, malaise, right upper quadrant pain, dark urine, pale stools, and jaundice. |