Details of Drug-Drug Interaction
| Drug General Information (ID: DDI7ML20V1) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Tetracosactide | Drug Info | Bacillus calmette-guerin substrain tice live antigen | Drug Info | |||||
| Drug Type | Small molecule | Vaccine | |||||||
| Therapeutic Class | Diagnostic Agents | Vaccine | |||||||
| Mechanism of Tetracosactide-Bacillus calmette-guerin substrain tice live antigen Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
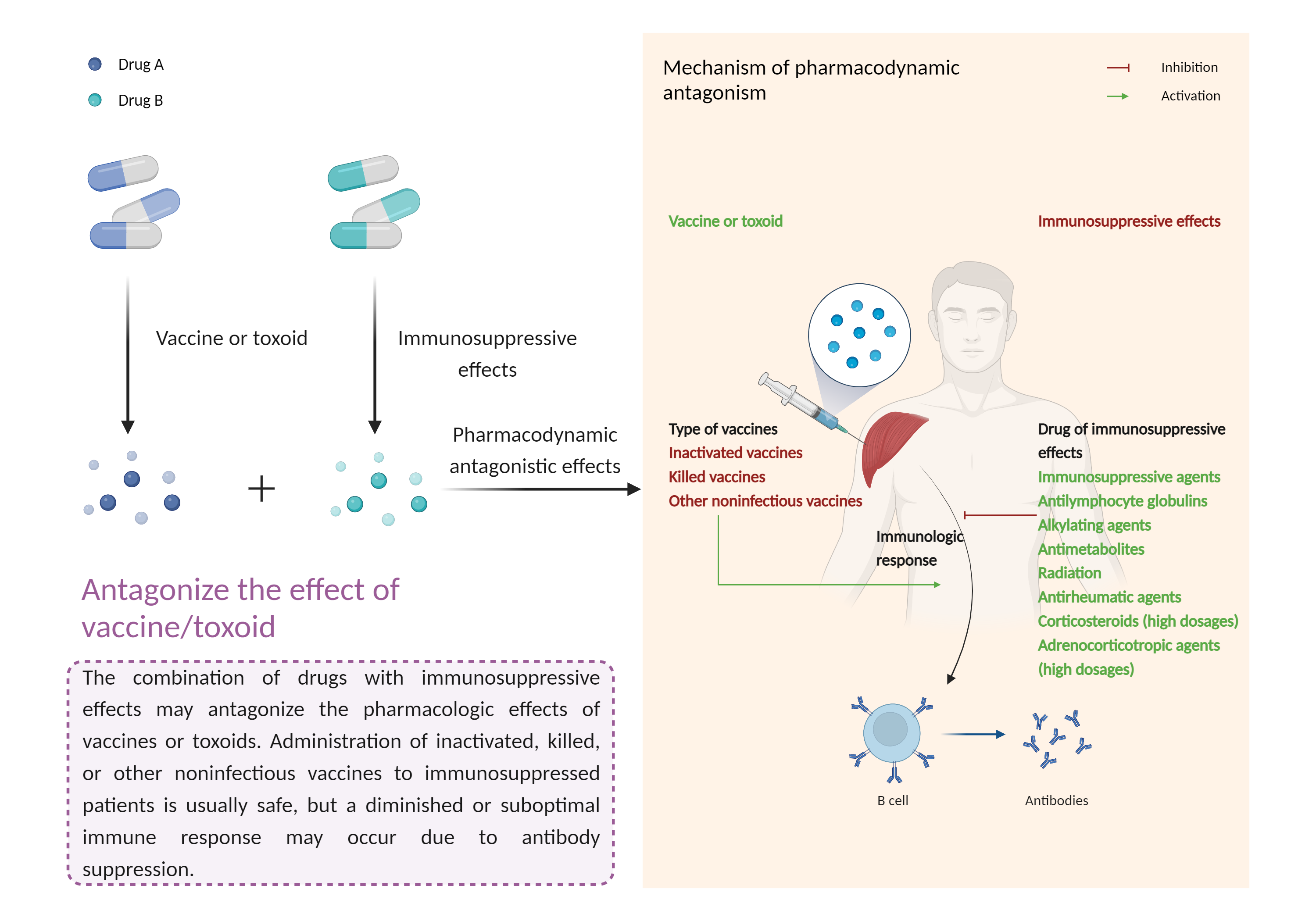
| Antagonize the effect of vaccine/toxoid Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Tetracosactide | Bacillus calmette-guerin substrain tice live antigen | |||||||
| Mechanism | Immunosuppressive effects | Vaccine or toxoid | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Vaccine/toxoid | ||||||||
| Factor Description | The beneficial effects of the vaccine/toxinoid may be reduced, leading to a diminished or suboptimal immune response. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | In general, live attenuated vaccines should not be used in patients receiving immunosuppressive corticosteroid therapy. Vaccination should be deferred until after such therapy is discontinued and immune function has been restored. The interval depends on the dosage and duration of corticosteroid therapy administered, but may be at least 3 months in most cases. Current local immunization guidelines should be consulted for recommendations. In patients who have recently been vaccinated, high-dose corticosteroid therapy should not be initiated for at least 2 weeks. Vaccines may generally be administered to patients receiving corticosteroids as replacement therapy (e.g., for Addison's disease). | ||||||||
