Details of Drug-Drug Interaction
| Drug General Information (ID: DDI6YJKVHW) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Nabumetone | Drug Info | Pitolisant | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Nsaids/Analgesics | Cns Stimulants | |||||||
| Structure | |||||||||
| Mechanism of Nabumetone-Pitolisant Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
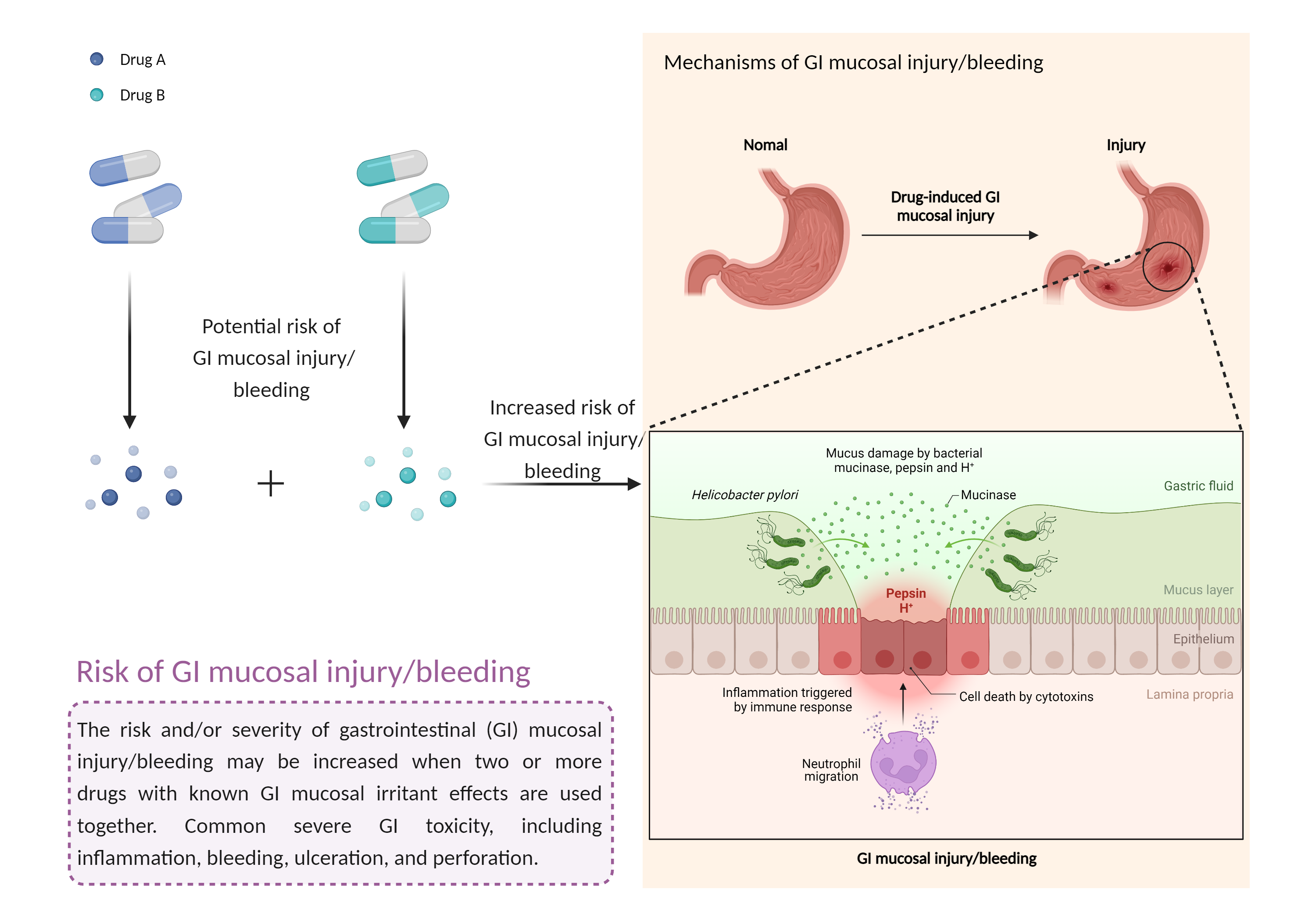
| Increased risk of GI mucosal injury/bleeding risk Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Nabumetone | Pitolisant | |||||||
| Mechanism | GI mucosal injury/bleeding risk | GI mucosal injury/bleeding risk | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Gastrointestinal toxicity | ||||||||
| Factor Description | The risk and severity of gastrointestinal toxicity, including inflammation, bleeding, ulceration, and perforation, increases when drugs with gastrointestinal mucosal irritant effects are combined. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if pitolisant is used in combination with NSAIDs and/or corticosteroids, particularly in patients with a prior history of peptic ulcer disease or GI bleeding and in elderly or debilitated patients. If concomitant therapy is required, patients should be advised to report signs and symptoms of adverse GI effects, including abdominal pain or discomfort, dyspepsia, gastroesophageal reflux disease, gastritis, or the appearance of black, tarry stools. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||||||||||||||||
