Details of Drug-Drug Interaction
| Drug General Information (ID: DDI5QVA192) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Naltrexone | Drug Info | Bremelanotide | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antidotes | Melanocortin Receptor Agonists | |||||||
| Structure | |||||||||
| Mechanism of Naltrexone-Bremelanotide Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
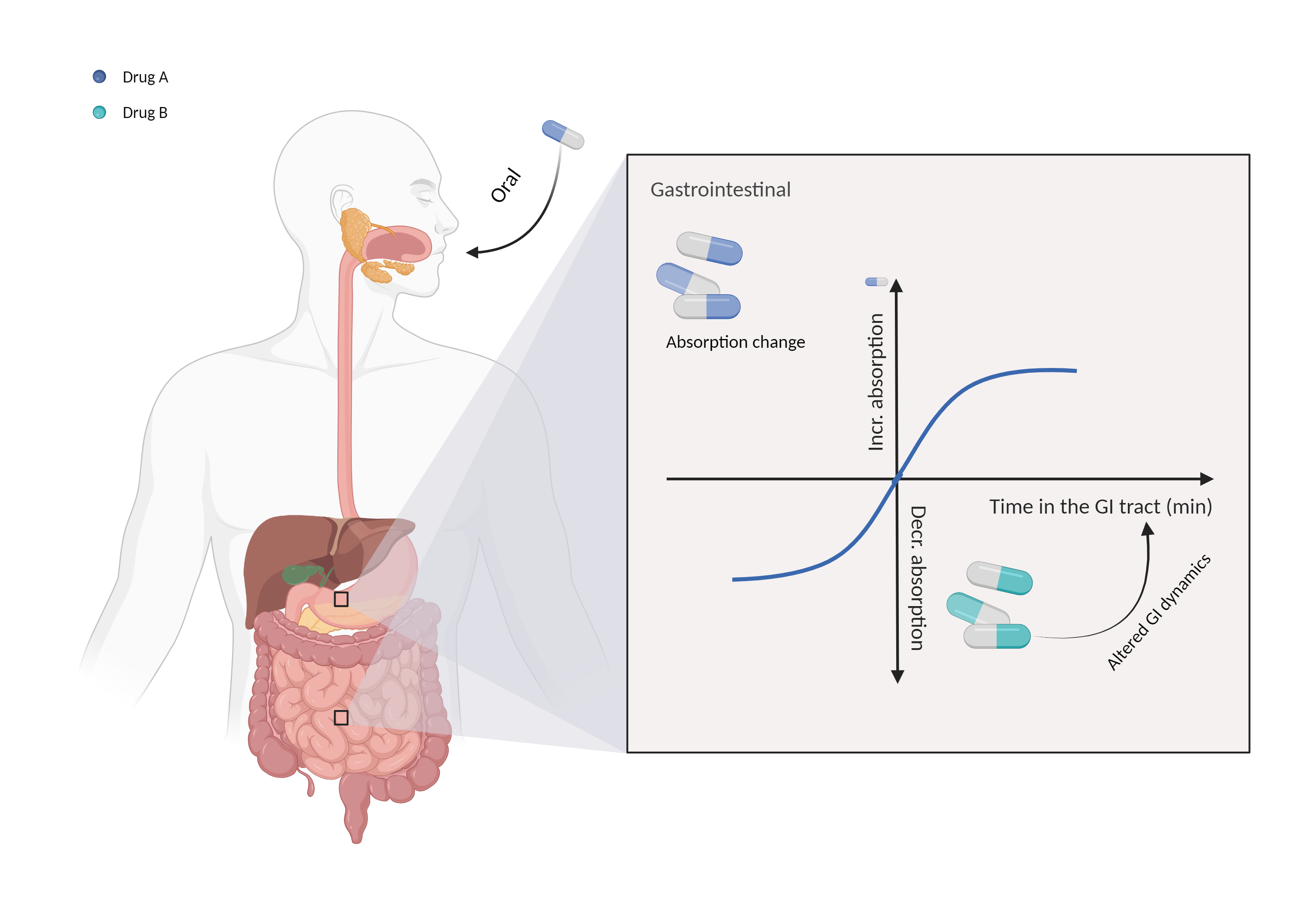
| Altered gastrointestinal dynamics Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Naltrexone | Bremelanotide | |||||||
| Mechanism | Absorption affected by delayed gastric emptying | Delayed gastric emptying | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Gastrointestinal Motility | ||||||||
| Factor Description | Gastrointestinal motility is an important factor in determining the absorption of orally administered drugs, and it controls the residence time of the drug in the digestive tract. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The prescribing information for bremelanotide recommends avoiding concomitant use with orally administered naltrexone-containing products that are intended for the treatment of alcohol or opioid addiction. When naltrexone is used for other indications, monitor for potentially reduced therapeutic efficacy when coadministered with bremelanotide. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Vyleesi (bremelanotide). AMAG Pharmaceuticals Inc, Lexington, MA. | ||||||||||||||||||
