Details of Drug-Drug Interaction
| Drug General Information (ID: DDI4MRA9XB) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Human C1-esterase inhibitor | Drug Info | Peginesatide | Drug Info | |||||
| Drug Type | Protein/peptide | Protein/peptide | |||||||
| Therapeutic Class | Hereditary Angioedema Agents | Recombinant Human Erythropoietins | |||||||
| Mechanism of Human C1-esterase inhibitor-Peginesatide Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
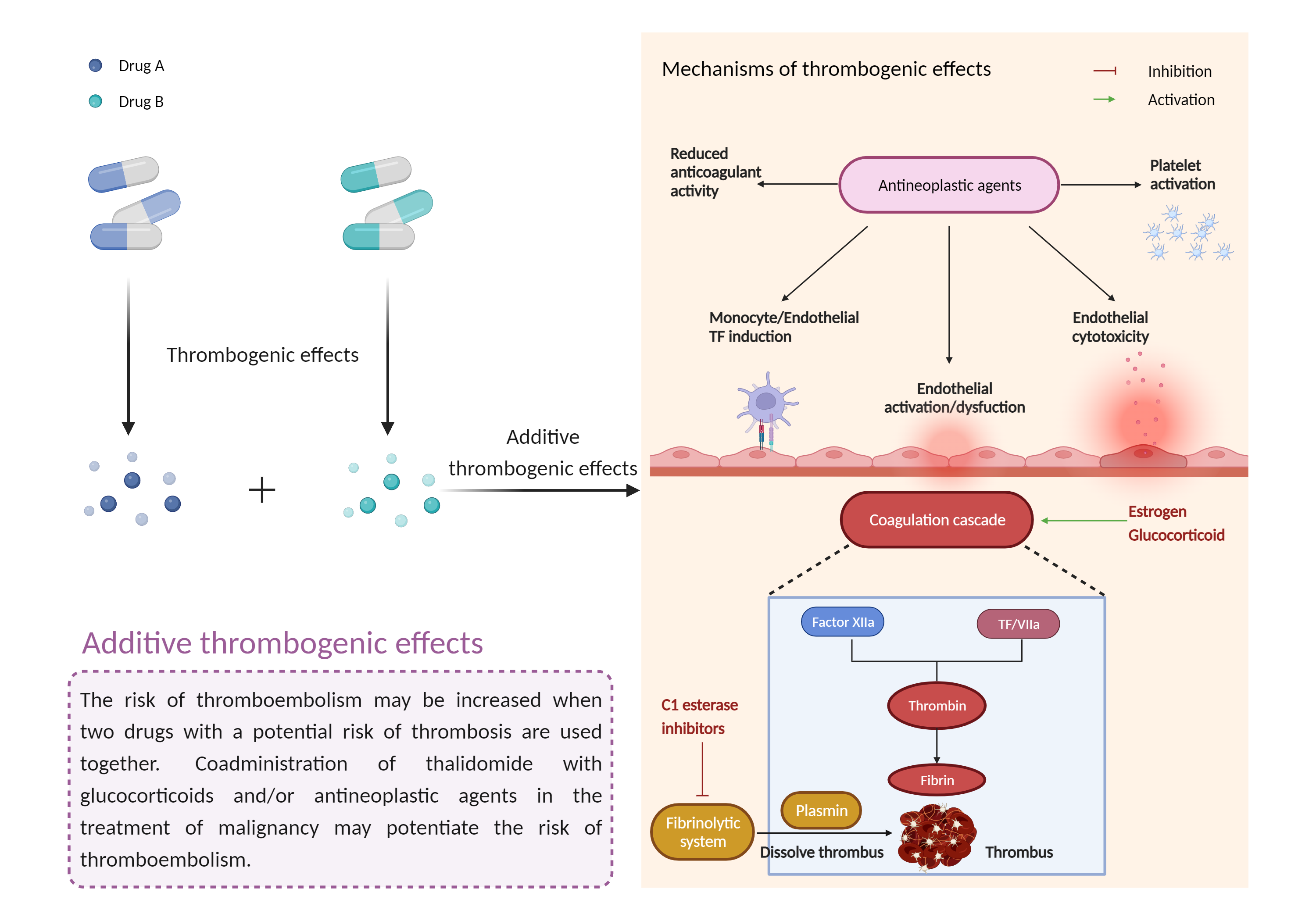
| Additive thrombogenic effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Human C1-esterase inhibitor | Peginesatide | |||||||
| Mechanism | Thrombogenic effects | Thrombogenic effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Thrombogenic effects | ||||||||
| Factor Description | Drug-induced thrombosis disrupts the balance between thrombogenic factors and protective mechanisms and tends to produce thrombosis. Drug-induced thrombosis usually causes venous thrombotic events, and arterial thrombotic events are also noted as a result of drug administration | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised when C1 esterase inhibitors are used with other drugs that have thrombogenic effects such as androgens, estrogens, erythropoiesis-stimulating agents, or selective estrogen receptor modulators. Close monitoring for thromboembolic events is recommended during and after administration of C1 esterase inhibitors. Patients should be advised to seek medical attention if they develop potential signs and symptoms of thromboembolism such as chest pain shortness of breath rapid pulse pain, swelling, and/or discoloration in an arm or leg and numbness or weakness on one side of the body. | ||||||||
