| Management |
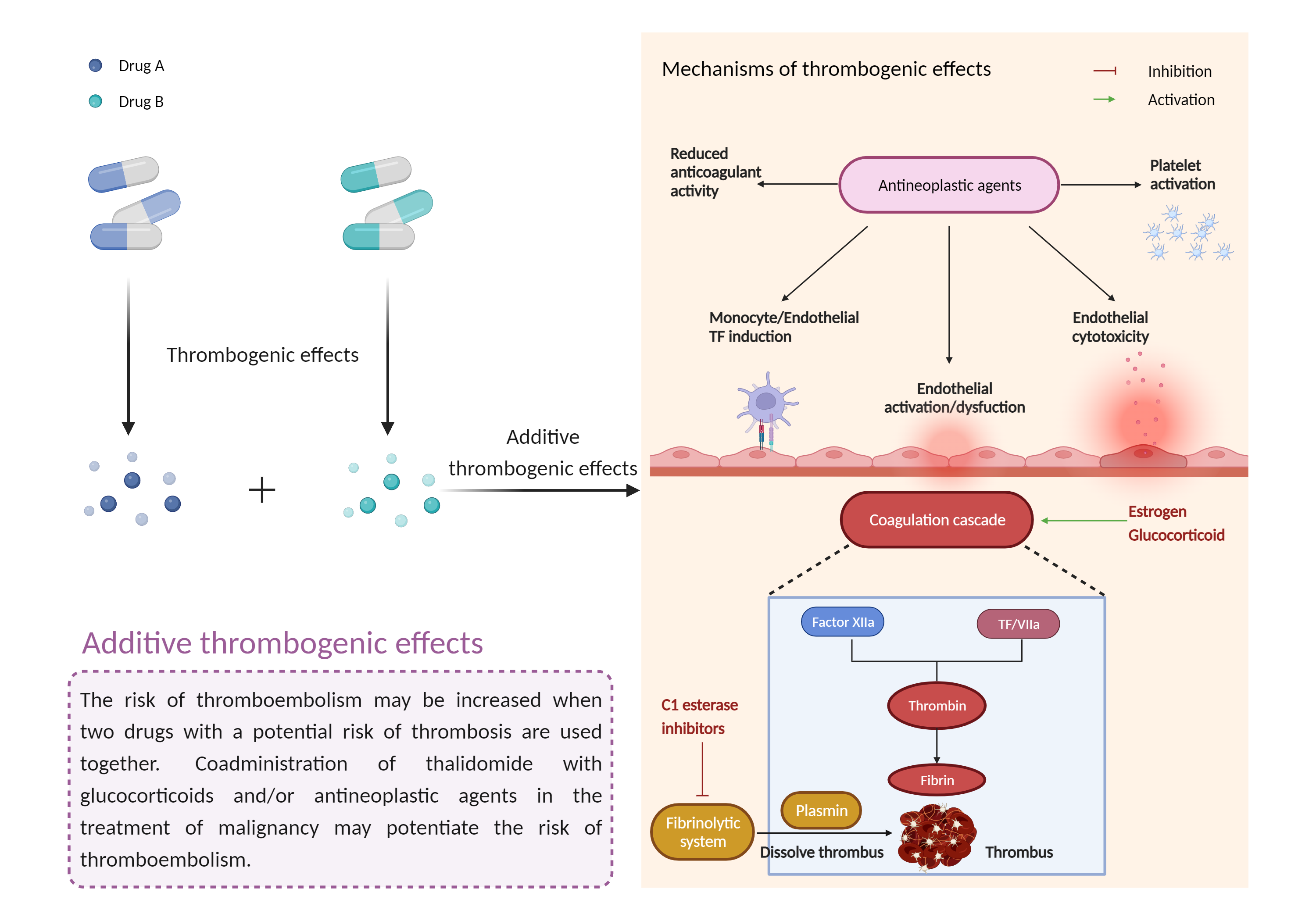
Agents that are known to cause thrombosis should be used with caution in multiple myeloma patients receiving thalidomide with standard chemotherapeutic agents and/or steroids. Patients should be advised to seek medical attention if they develop potential signs and symptoms of thromboembolism such as chest pain shortness of breath rapid pulse pain, swelling, and/or discoloration in an arm or leg and numbness or weakness on one side of the body. Prophylaxis with anticoagulants such as low-molecular weight heparins or warfarin may be appropriate, but the decision to take thromboprophylactic measures should be made after careful assessment of underlying risk factors. If a thromboembolic event occurs during therapy with thalidomide, treatment must be discontinued and standard anticoagulation therapy initiated. Once anticoagulation is stabilized and complications of the thromboembolic event under control, thalidomide may be restarted at the original dose if benefit is deemed to outweigh the risks. Anticoagulation therapy should be continued during the remaining course of thalidomide treatment. |