Details of Drug-Drug Interaction
| Drug General Information (ID: DDI3CLOV4A) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Insulin lispro | Drug Info | Lixisenatide | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antidiabetic Agents | Antidiabetic Agents | |||||||
| Structure | |||||||||
| Mechanism of Insulin lispro-Lixisenatide Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
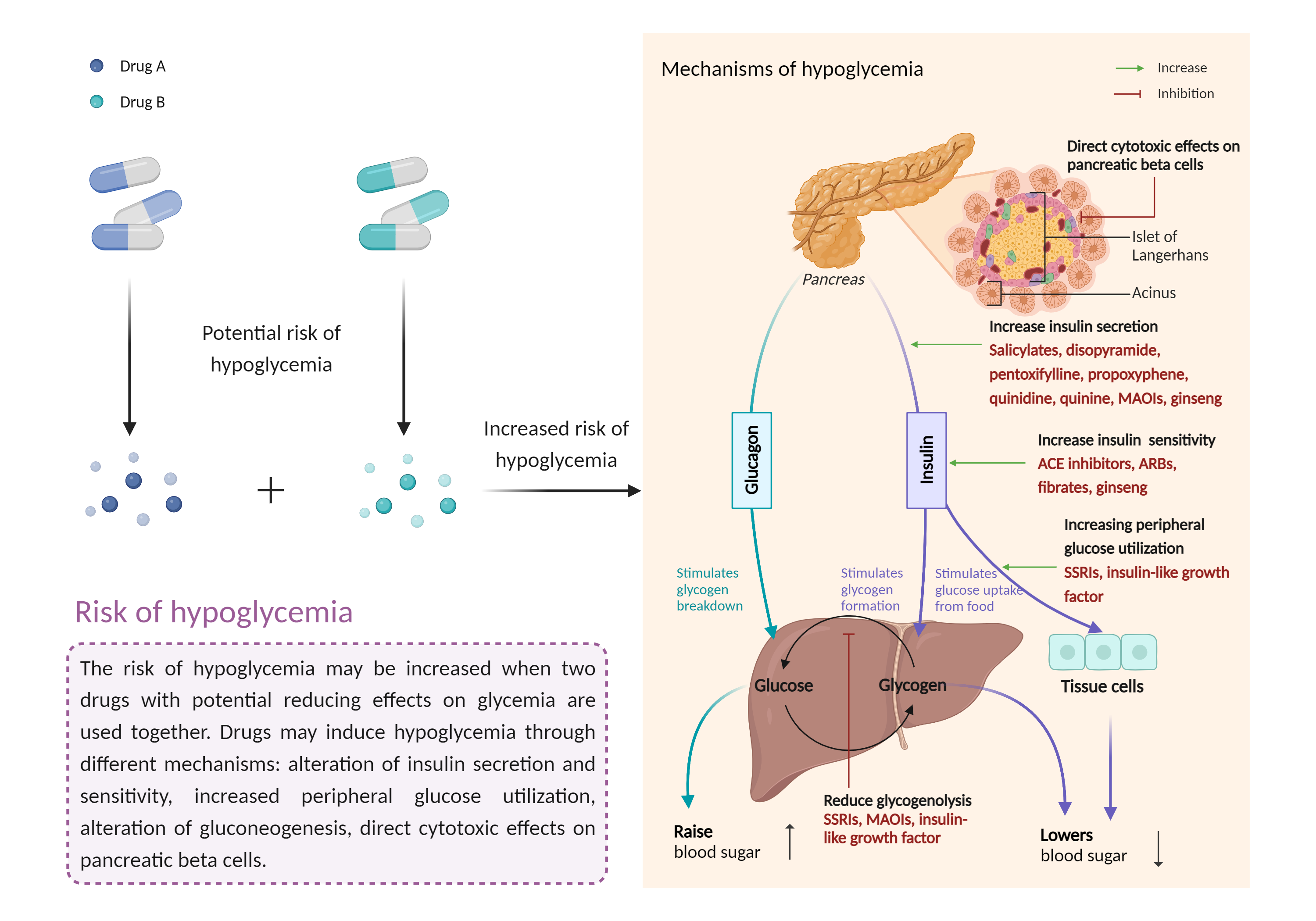
| Increased risk of hypoglycemia Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Insulin lispro | Lixisenatide | |||||||
| Mechanism | Hypoglycemic effects | Hypoglycemic effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hypoglycemia | ||||||||
| Factor Description | Hypoglycemia is a condition in which your blood sugar (glucose) level is below the standard range. It causes irregular or rapid heartbeat, pale skin, numbness of the lips, tongue or cheeks, and sweating. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | When a GLP-1 receptor agonist is used as add-on therapy to basal insulin, a lower dosage of insulin may be required. Blood glucose should be monitored closely, and patients should be counseled to recognize the symptoms of hypoglycemia such as headache, dizziness, drowsiness, nervousness, confusion, tremor, hunger, weakness, perspiration, palpitation, and tachycardia. If hypoglycemia occurs, patients should initiate appropriate remedial therapy immediately and contact their physician. Patients should also be advised to take precautions to avoid hypoglycemia while driving or operating hazardous machinery. Concurrent use of GLP-1 receptor agonists with prandial insulin has not been studied and is generally not recommended. | ||||||||
