Details of Drug-Drug Interaction
| Drug General Information (ID: DDI3B6IDK5) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Cyclosporine | Drug Info | Valsartan | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiviral Agents | Antihypertensive Agents | |||||||
| Structure | |||||||||
| Mechanism of Cyclosporine-Valsartan Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
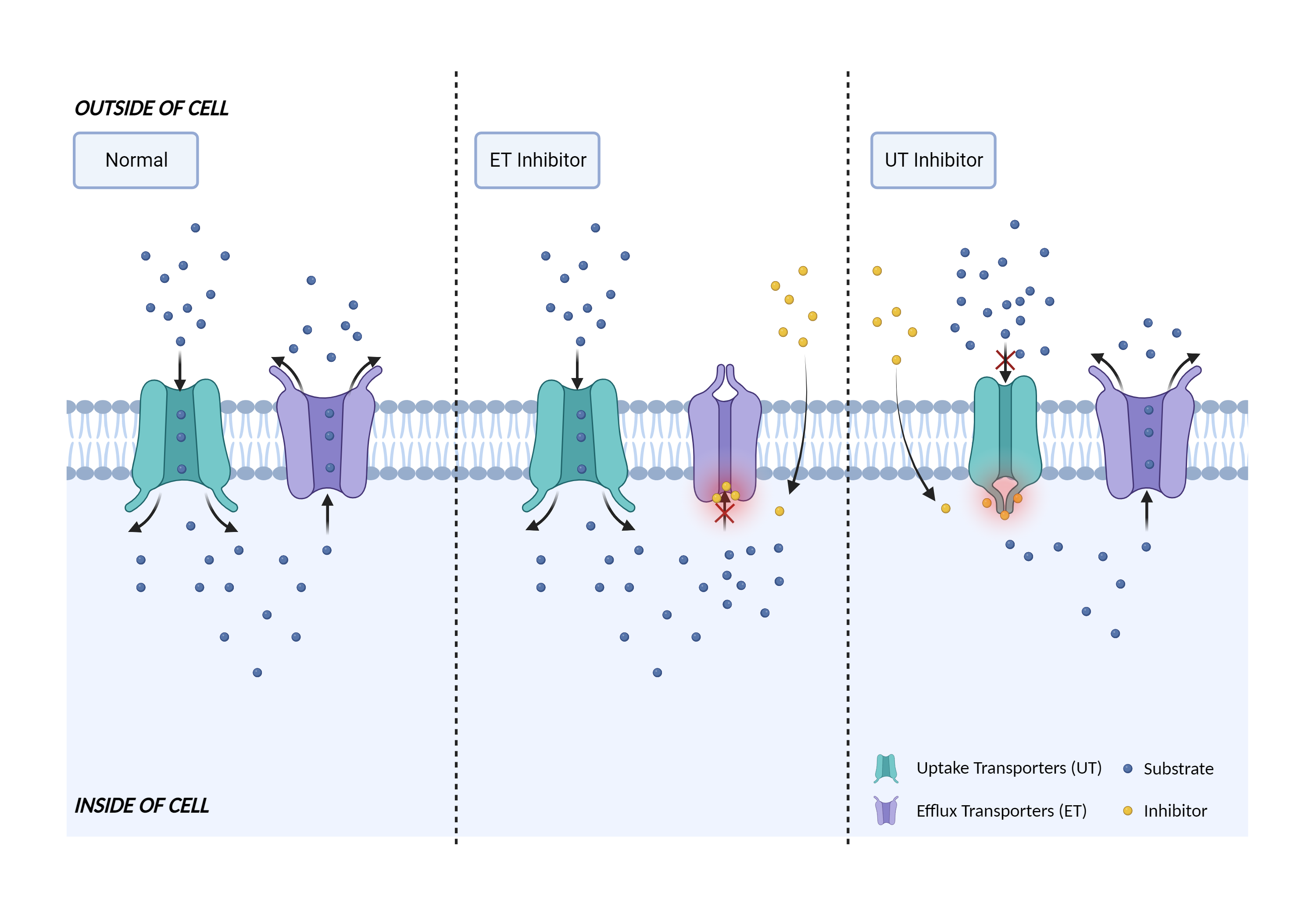
| Transporter inhibition Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Cyclosporine | Valsartan | |||||||
| Mechanism 1 | OATP1B1 inhibitor | OATP1B1 substrate | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Liver organic anion transporter 1 |
×
Structure
Sequence
MDQNQHLNKTAEAQPSENKKTRYCNGLKMFLAALSLSFIAKTLGAIIMKSSIIHIERRFEISSSLVGFIDGSFEIGNLLVIVFVSYFGSKLHRPKLIGIGCFIMGIGGVLTALPHFFMGYYRYSKETNINSSENSTSTLSTCLINQILSLNRASPEIVGKGCLKESGSYMWIYVFMGNMLRGIGETPIVPLGLSYIDDFAKEGHSSLYLGILNAIAMIGPIIGFTLGSLFSKMYVDIGYVDLSTIRITPTDSRWVGAWWLNFLVSGLFSIISSIPFFFLPQTPNKPQKERKASLSLHVLETNDEKDQTANLTNQGKNITKNVTGFFQSFKSILTNPLYVMFVLLTLLQVSSYIGAFTYVFKYVEQQYGQPSSKANILLGVITIPIFASGMFLGGYIIKKFKLNTVGIAKFSCFTAVMSLSFYLLYFFILCENKSVAGLTMTYDGNNPVTSHRDVPLSYCNSDCNCDESQWEPVCGNNGITYISPCLAGCKSSSGNKKPIVFYNCSCLEVTGLQNRNYSAHLGECPRDDACTRKFYFFVAIQVLNLFFSALGGTSHVMLIVKIVQPELKSLALGFHSMVIRALGGILAPIYFGALIDTTCIKWSTNNCGTRGSCRTYNSTSFSRVYLGLSSMLRVSSLVLYIILIYAMKKKYQEKDINASENGSVMDEANLESLNKNKHFVPSAGADSETHC
|
|||||||
| Gene Name | OATP1B1 | ||||||||
| Uniprot ID | SO1B1_HUMAN | ||||||||
| KEGG Pathway | hsa:10599 | ||||||||
| Protein Family | Organo anion transporter (TC 2.A.60) family | ||||||||
| Protein Function |
Mediates the Na(+)-independent uptake of organic anions such as pravastatin, taurocholate, methotrexate, dehydroepiandrosterone sulfate, 17-beta-glucuronosyl estradiol, estrone sulfate, prostaglandin E2, thromboxane B2, leukotriene C3, leukotriene E4, thyroxine and triiodothyronine. Involved in the clearance of bile acids and organic anions from the liver.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
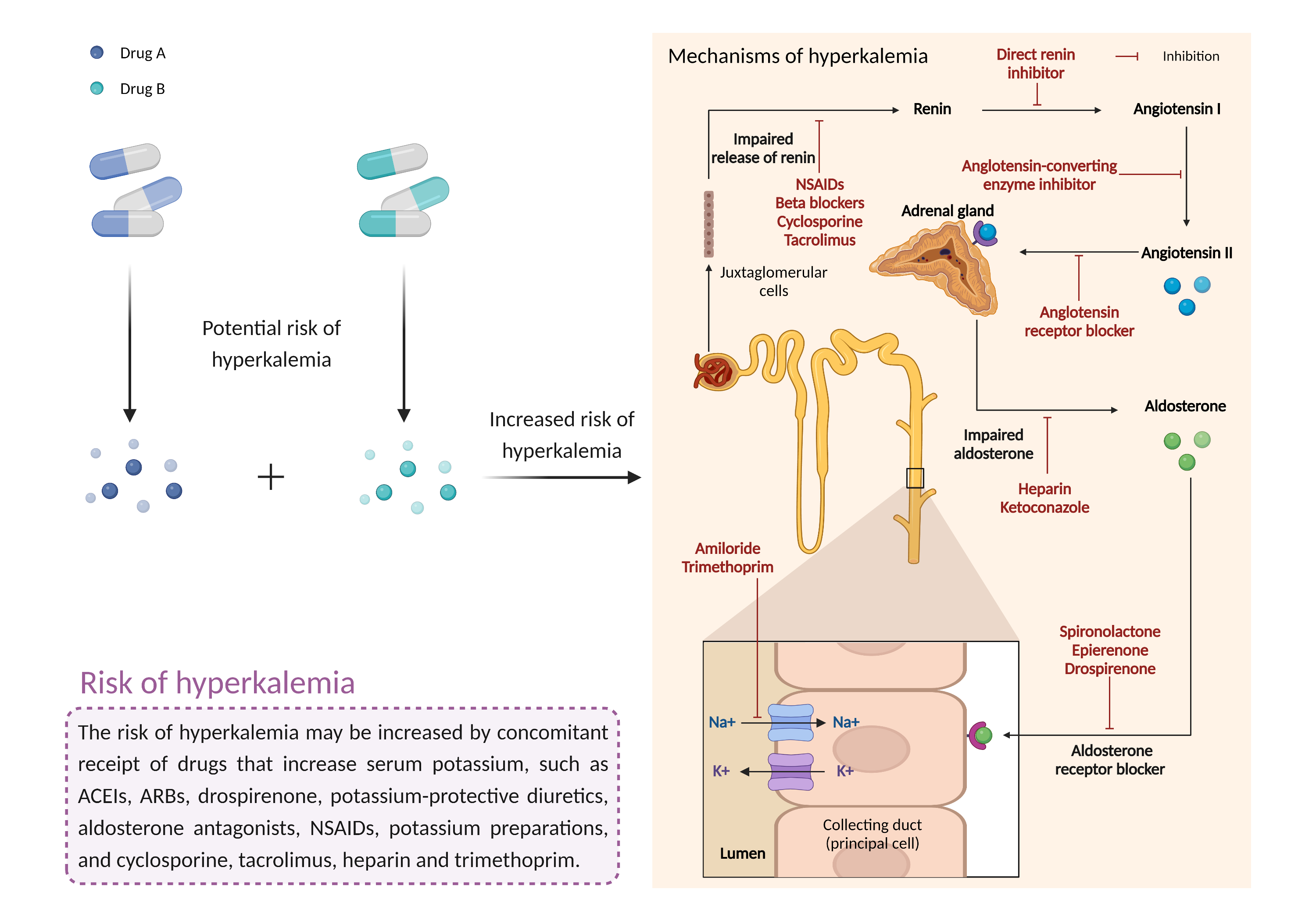
| Increased risk of hyperkalemia Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Cyclosporine | Valsartan | |||||||
| Mechanism 2 | Hyperkalemia | Hyperkalemia | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Hyperkalemia | ||||||||
| Factor Description | Hyperkalemia is a condition in which the level of potassium in the blood is higher than normal. While mild cases may not produce symptoms, severe hyperkalemia can lead to fatal arrhythmias if left untreated. | ||||||||
| Mechanism Description |
|
||||||||
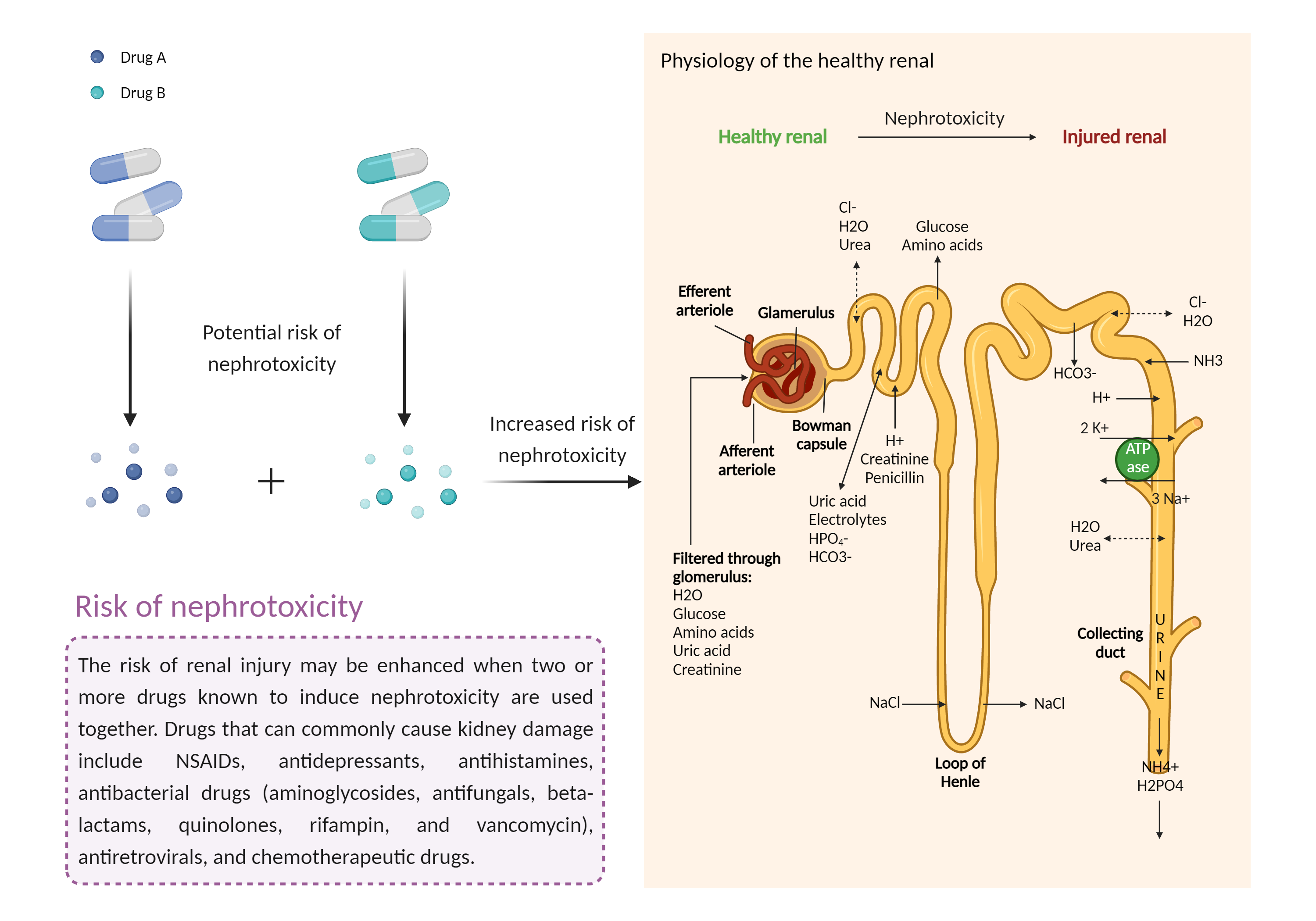
| Increased risk of nephrotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Cyclosporine | Valsartan | |||||||
| Mechanism 3 | Nephrotoxicity | Nephrotoxicity | |||||||
| Key Mechanism Factor 3 | |||||||||
| Factor Name | Nephrotoxicity | ||||||||
| Factor Description | The combination of drugs that can induce nephrotoxicity may increase the risk of kidney injury. When kidney injury occurs, the inability to remove excess urine and waste from the body can lead to high levels of urea nitrogen, creatinine, and electrolytes (such as potassium and magnesium) in the blood. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if valsartan is used in combination with cyclosporine. Pharmacologic response and blood pressure should be monitored more closely following the addition, discontinuation, or change of dosage of cyclosporine, and the valsartan regimen adjusted as necessary. Serum potassium levels and renal function should also be checked regularly. Particular caution is warranted in patients with renal impairment, diabetes, old age, severe or worsening heart failure, or dehydration. Patients should be advised to seek medical attention if they experience signs and symptoms of hyperkalemia such as nausea, vomiting, weakness, listlessness, tingling of the extremities, paralysis, confusion, weak pulse, and a slow or irregular heartbeat. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Diovan (valsartan). Novartis Pharmaceuticals, East Hanover, NJ. | ||||||||||||||||||
| 2 | Product Information. Exforge (amlodipine-valsartan). Novartis Pharmaceuticals, East Hanover, NJ. | ||||||||||||||||||
