| Mechanism of Cyclosporine-Valganciclovir Interaction
(Severity Level: Moderate)
|
|
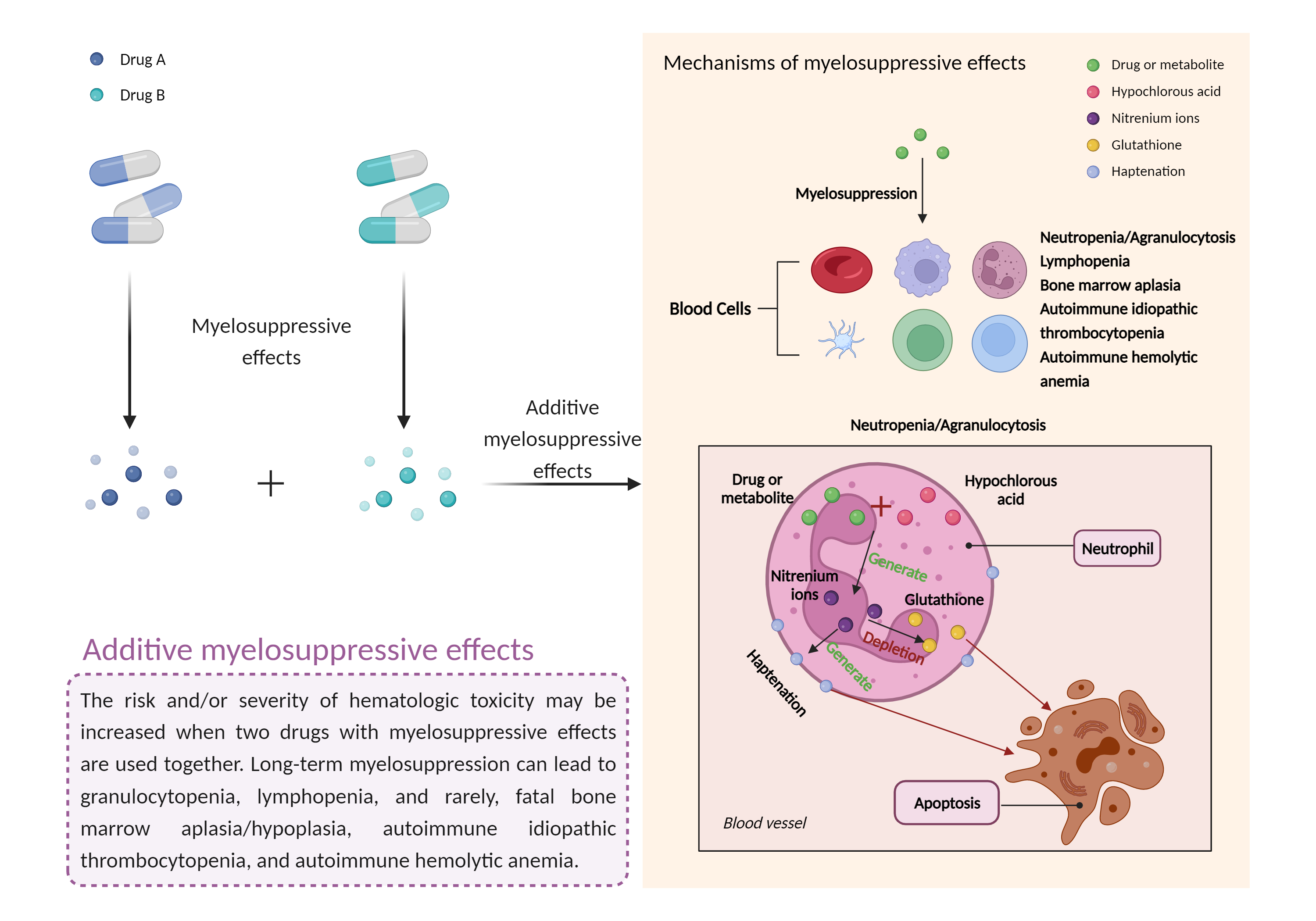
Additive myelosuppressive effects
Click to Show/Hide Mechanism Graph
|
 |
| Drug Name |
Cyclosporine |
Valganciclovir |
|
Mechanism 1
|
Myelosuppressive effects |
Myelosuppressive effects |
| Key Mechanism Factor 1 |
| Factor Name |
Myelosuppressive effects |
| Factor Description |
Myelosuppression, also known as bone marrow suppression, is a decrease in bone marrow activity that leads to a decrease in the production of blood cells. Some blood cell disorders include: erythrocytopenia (anemia), leukopenia (neutropenia), and thrombocytopenia (thrombocytopenia). |
| Mechanism Description |
- Additive myelosuppressive effects by the combination of Cyclosporine and Valganciclovir
|
|
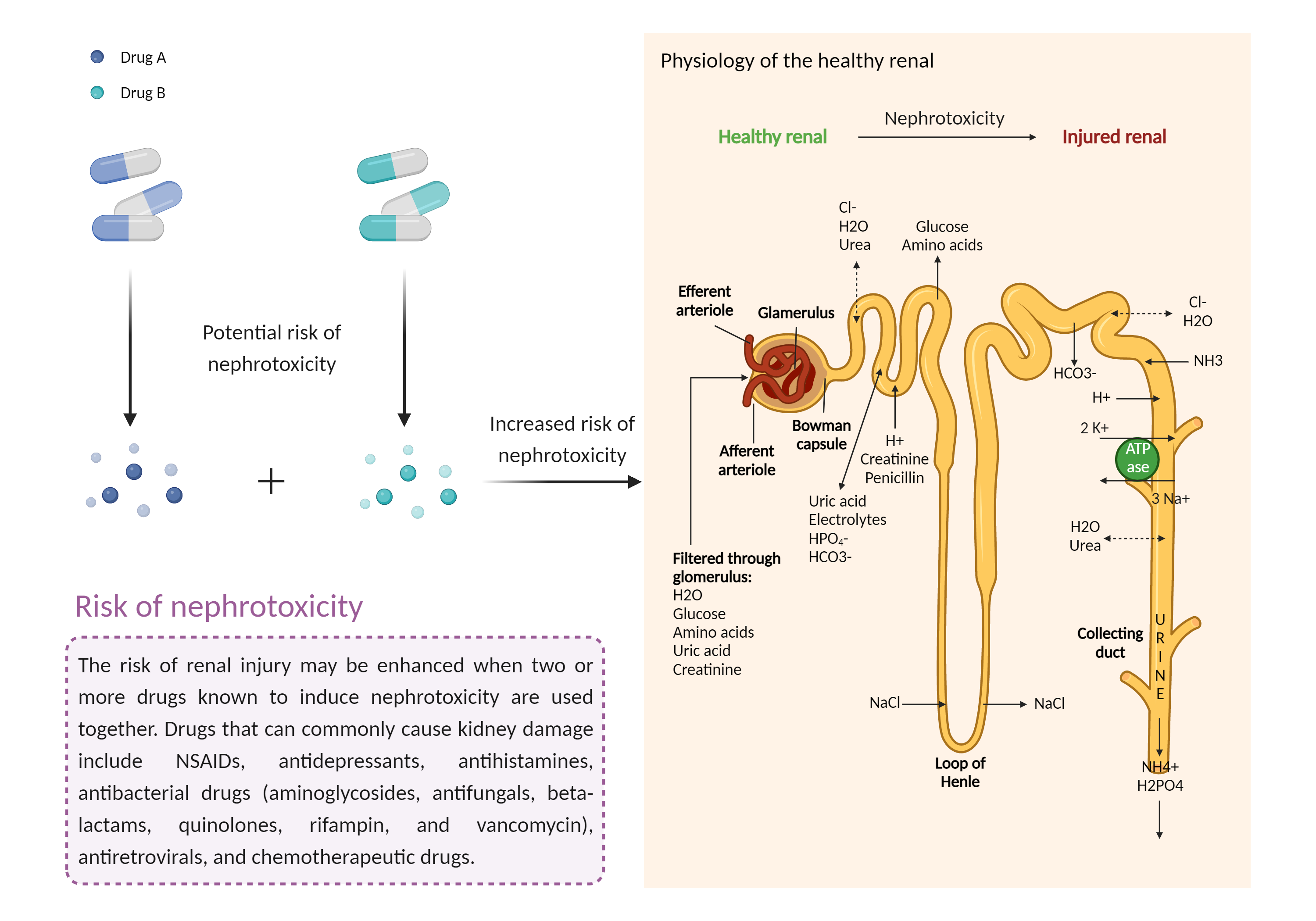
Increased risk of nephrotoxicity
Click to Show/Hide Mechanism Graph
|
 |
| Drug Name |
Cyclosporine |
Valganciclovir |
|
Mechanism 2
|
Nephrotoxicity |
Nephrotoxicity |
| Key Mechanism Factor 2 |
| Factor Name |
Nephrotoxicity |
| Factor Description |
The combination of drugs that can induce nephrotoxicity may increase the risk of kidney injury. When kidney injury occurs, the inability to remove excess urine and waste from the body can lead to high levels of urea nitrogen, creatinine, and electrolytes (such as potassium and magnesium) in the blood. |
| Mechanism Description |
- Increased risk of nephrotoxicity by the combination of Cyclosporine and Valganciclovir
|


