Details of Drug-Drug Interaction
| Drug General Information (ID: DDI26ZUSK0) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Digoxin | Drug Info | Dexlansoprazole | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiarrhythmic Agents | Proton Pump Inhibitors | |||||||
| Structure | |||||||||
| Mechanism of Digoxin-Dexlansoprazole Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
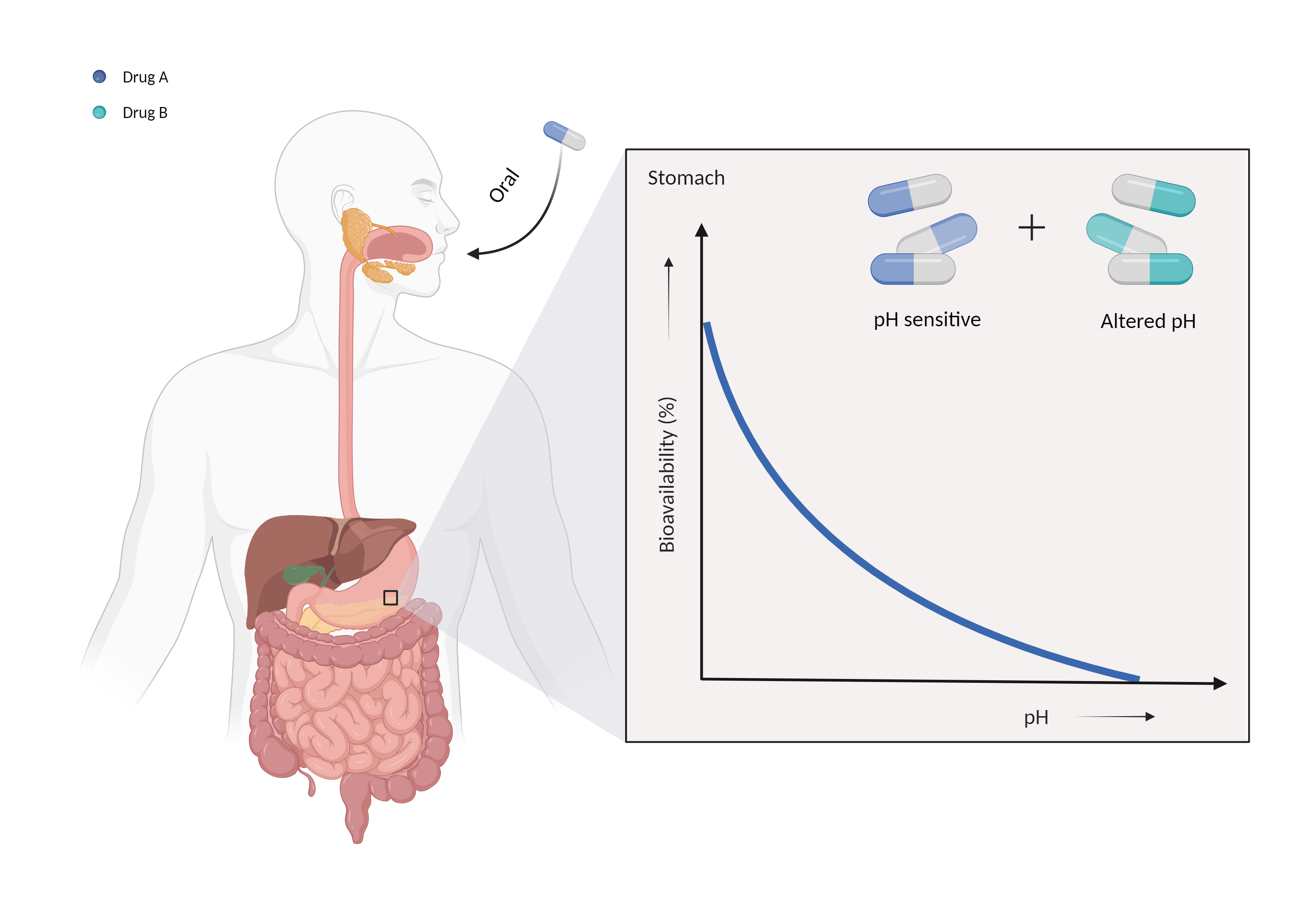
| Altered gastric pH Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Digoxin | Dexlansoprazole | |||||||
| Mechanism 1 | Gastric alkalinizer | Gastric pH sensitive | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Gastric pH | ||||||||
| Factor Description | The normal pH range of gastric acid is between 1.5 and 3.5 and is highly acidic, consisting mainly of hydrochloric acid. Changes in the pH of the stomach can alter the absorption of drugs. | ||||||||
| Mechanism Description |
|
||||||||
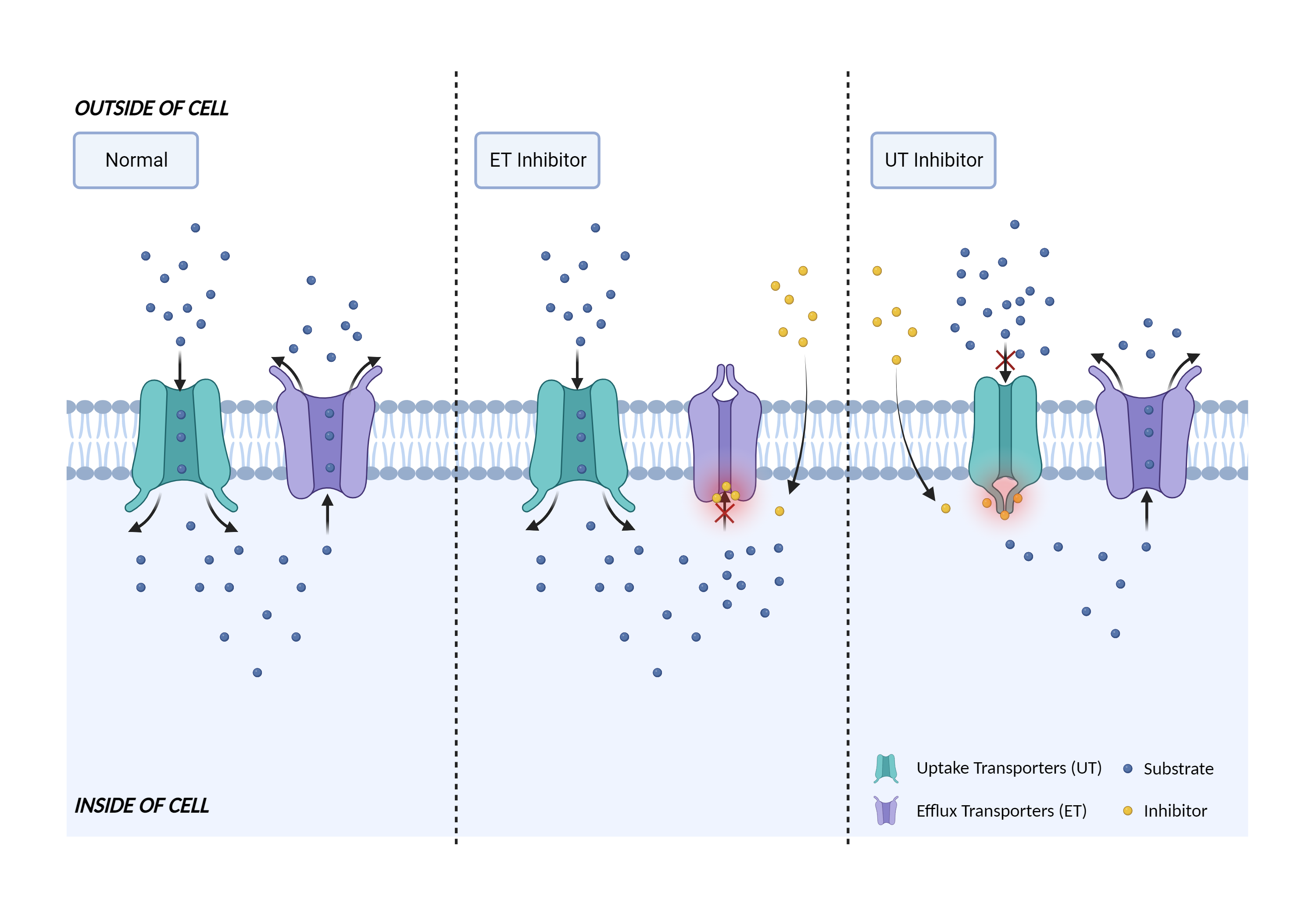
| Transporter inhibition Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Digoxin | Dexlansoprazole | |||||||
| Mechanism 2 | P-gp substrate | P-gp inhibitor | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | P-glycoprotein 1 |
×
Structure
Sequence
MDLEGDRNGGAKKKNFFKLNNKSEKDKKEKKPTVSVFSMFRYSNWLDKLYMVVGTLAAIIHGAGLPLMMLVFGEMTDIFANAGNLEDLMSNITNRSDINDTGFFMNLEEDMTRYAYYYSGIGAGVLVAAYIQVSFWCLAAGRQIHKIRKQFFHAIMRQEIGWFDVHDVGELNTRLTDDVSKINEGIGDKIGMFFQSMATFFTGFIVGFTRGWKLTLVILAISPVLGLSAAVWAKILSSFTDKELLAYAKAGAVAEEVLAAIRTVIAFGGQKKELERYNKNLEEAKRIGIKKAITANISIGAAFLLIYASYALAFWYGTTLVLSGEYSIGQVLTVFFSVLIGAFSVGQASPSIEAFANARGAAYEIFKIIDNKPSIDSYSKSGHKPDNIKGNLEFRNVHFSYPSRKEVKILKGLNLKVQSGQTVALVGNSGCGKSTTVQLMQRLYDPTEGMVSVDGQDIRTINVRFLREIIGVVSQEPVLFATTIAENIRYGRENVTMDEIEKAVKEANAYDFIMKLPHKFDTLVGERGAQLSGGQKQRIAIARALVRNPKILLLDEATSALDTESEAVVQVALDKARKGRTTIVIAHRLSTVRNADVIAGFDDGVIVEKGNHDELMKEKGIYFKLVTMQTAGNEVELENAADESKSEIDALEMSSNDSRSSLIRKRSTRRSVRGSQAQDRKLSTKEALDESIPPVSFWRIMKLNLTEWPYFVVGVFCAIINGGLQPAFAIIFSKIIGVFTRIDDPETKRQNSNLFSLLFLALGIISFITFFLQGFTFGKAGEILTKRLRYMVFRSMLRQDVSWFDDPKNTTGALTTRLANDAAQVKGAIGSRLAVITQNIANLGTGIIISFIYGWQLTLLLLAIVPIIAIAGVVEMKMLSGQALKDKKELEGSGKIATEAIENFRTVVSLTQEQKFEHMYAQSLQVPYRNSLRKAHIFGITFSFTQAMMYFSYAGCFRFGAYLVAHKLMSFEDVLLVFSAVVFGAMAVGQVSSFAPDYAKAKISAAHIIMIIEKTPLIDSYSTEGLMPNTLEGNVTFGEVVFNYPTRPDIPVLQGLSLEVKKGQTLALVGSSGCGKSTVVQLLERFYDPLAGKVLLDGKEIKRLNVQWLRAHLGIVSQEPILFDCSIAENIAYGDNSRVVSQEEIVRAAKEANIHAFIESLPNKYSTKVGDKGTQLSGGQKQRIAIARALVRQPHILLLDEATSALDTESEKVVQEALDKAREGRTCIVIAHRLSTIQNADLIVVFQNGRVKEHGTHQQLLAQKGIYFSMVSVQAGTKRQ
|
|||||||
| Gene Name | ABCB1 | ||||||||
| Uniprot ID | MDR1_HUMAN | ||||||||
| KEGG Pathway | hsa:5243 | ||||||||
| Protein Family | ABC transporter superfamily | ||||||||
| Protein Function |
Translocates drugs and phospholipids across the membrane (PubMed:8898203, PubMed:2897240, PubMed:9038218). Catalyzes the flop of phospholipids from the cytoplasmic to the exoplasmic leaflet of the apical membrane. Participates mainly to the flop of phosphatidylcholine, phosphatidylethanolamine, beta-D-glucosylceramides and sphingomyelins (PubMed:8898203). Energy-dependent efflux pump responsible for decreased drug accumulation in multidrug-resistant cells (PubMed:2897240, PubMed:9038218).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
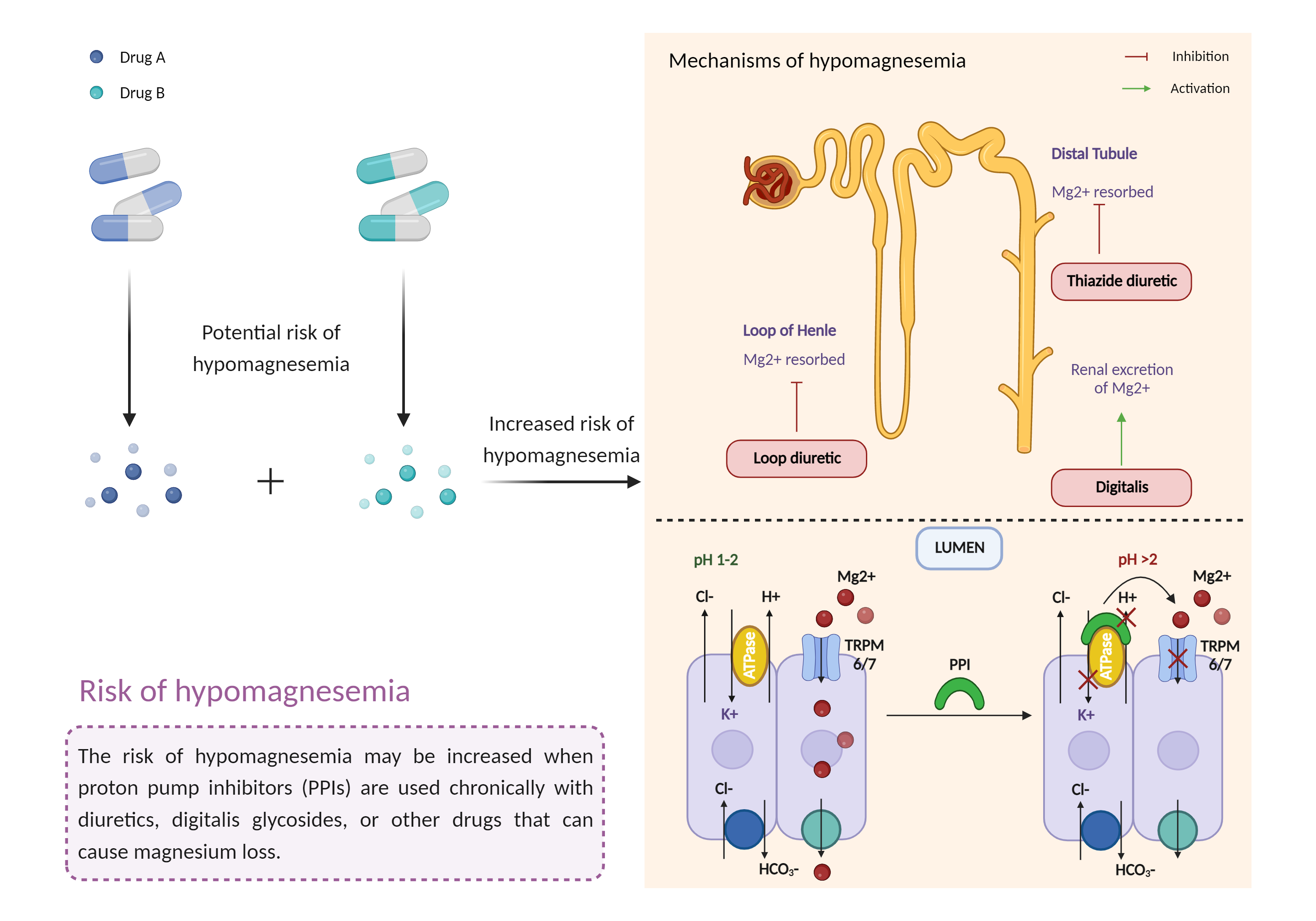
| Increased risk of hypomagnesemia Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Digoxin | Dexlansoprazole | |||||||
| Mechanism 3 | Hypomagnesemia | Hypomagnesemia | |||||||
| Key Mechanism Factor 3 | |||||||||
| Factor Name | Hypomagnesemia | ||||||||
| Factor Description | Hypomagnesemia is a condition that occurs when you have too much magnesium in your body. Symptoms of hypomagnesemia include: nausea, vomiting, neurological damage, abnormally low blood pressure (hypotension), flushing, and headaches. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if digoxin is prescribed in combination with PPIs. Pharmacologic response and serum digoxin levels should be monitored more closely following the addition or discontinuation of PPI therapy, and the digoxin dosage adjusted as necessary. Patients should be advised to notify their physician if they experience potential signs and symptoms of digoxin toxicity such as nausea, anorexia, visual disturbances, slow pulse, or irregular heartbeats. Monitoring of serum magnesium levels is recommended prior to initiation of PPI therapy and periodically thereafter if prolonged treatment is anticipated or when combined with other agents that can cause hypomagnesemia (e.g., diuretics, aminoglycosides, cation exchange resins, amphotericin B, cetuximab, cisplatin, cyclosporine, foscarnet, panitumumab, pentamidine, tacrolimus). Patients should be advised to seek immediate medical attention if they develop potential signs and symptoms of hypomagnesemia such as palpitations, arrhythmia, muscle spasm, tremor, or convulsions. In children, abnormal heart rates may cause fatigue, upset stomach, dizziness, and lightheadedness. Magnesium replacement as well as discontinuation of the PPI may be required in some patients. | ||||||||
