Details of Drug-Drug Interaction
| Drug General Information (ID: DDI21N3ACO) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Tobramycin | Drug Info | Neostigmine | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antibiotics | Parasympathomimetics | |||||||
| Structure | |||||||||
| Mechanism of Tobramycin-Neostigmine Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
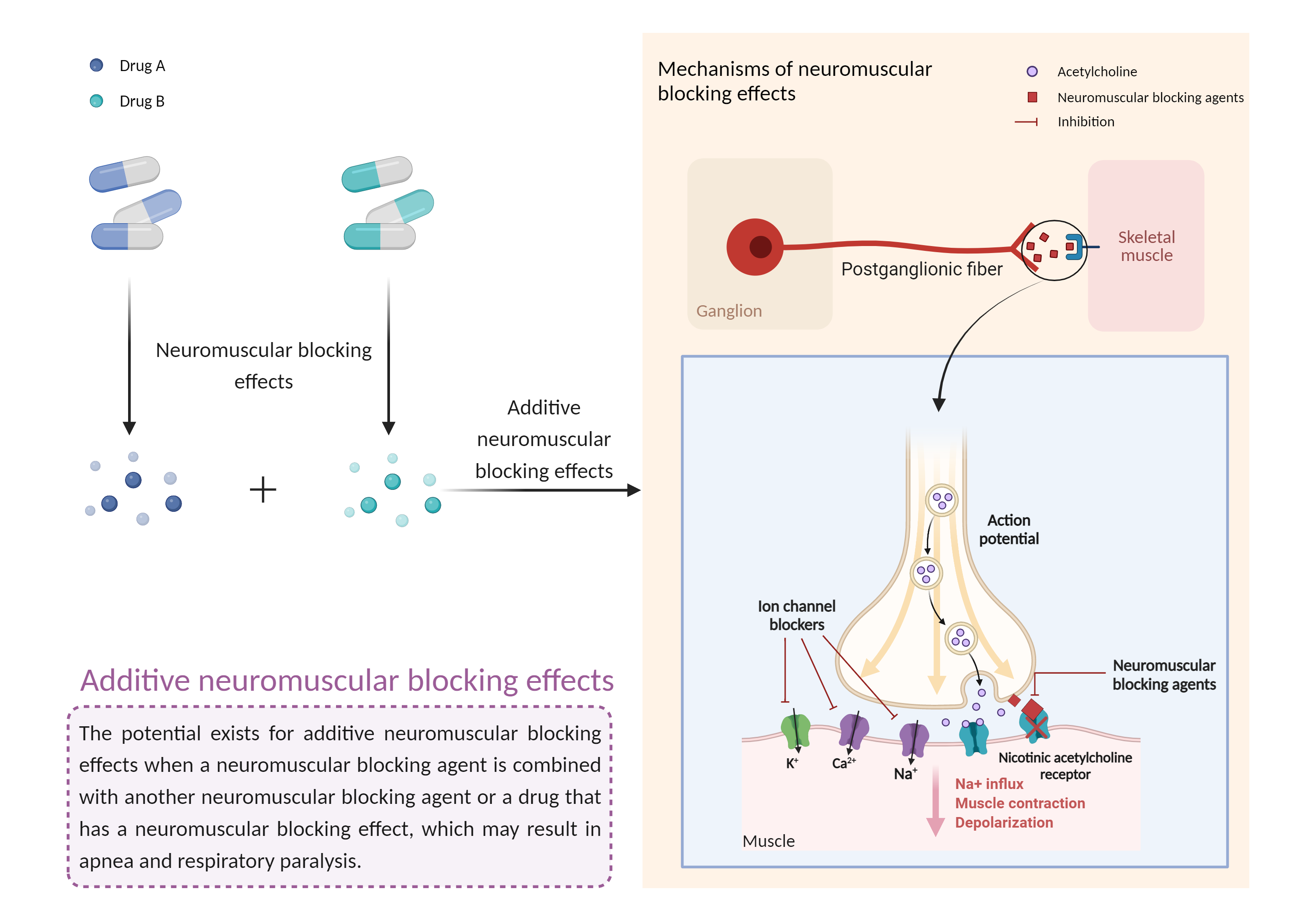
| Additive neuromuscular blocking effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Tobramycin | Neostigmine | |||||||
| Mechanism |
Neuromuscular blocking effects Aminoglycosides |
Neuromuscular blocking effects Acetylcholinesterase Inhibitor |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Acetylcholinesterase |
×
Structure
Sequence
MRPPQCLLHTPSLASPLLLLLLWLLGGGVGAEGREDAELLVTVRGGRLRGIRLKTPGGPVSAFLGIPFAEPPMGPRRFLPPEPKQPWSGVVDATTFQSVCYQYVDTLYPGFEGTEMWNPNRELSEDCLYLNVWTPYPRPTSPTPVLVWIYGGGFYSGASSLDVYDGRFLVQAERTVLVSMNYRVGAFGFLALPGSREAPGNVGLLDQRLALQWVQENVAAFGGDPTSVTLFGESAGAASVGMHLLSPPSRGLFHRAVLQSGAPNGPWATVGMGEARRRATQLAHLVGCPPGGTGGNDTELVACLRTRPAQVLVNHEWHVLPQESVFRFSFVPVVDGDFLSDTPEALINAGDFHGLQVLVGVVKDEGSYFLVYGAPGFSKDNESLISRAEFLAGVRVGVPQVSDLAAEAVVLHYTDWLHPEDPARLREALSDVVGDHNVVCPVAQLAGRLAAQGARVYAYVFEHRASTLSWPLWMGVPHGYEIEFIFGIPLDPSRNYTAEEKIFAQRLMRYWANFARTGDPNEPRDPKAPQWPPYTAGAQQYVSLDLRPLEVRRGLRAQACAFWNRFLPKLLSATDTLDEAERQWKAEFHRWSSYMVHWKNQFDHYSKQDRCSDL
|
|||||||
| Gene Name | ACHE | ||||||||
| Uniprot ID | ACES_HUMAN | ||||||||
| KEGG Pathway | hsa:43 | ||||||||
| Protein Family | Type-B carboxylesterase/lipase family | ||||||||
| Protein Function |
Hydrolyzes rapidly the acetylcholine neurotransmitter released into the synaptic cleft allowing to terminate the signal transduction at the neuromuscular junction. Role in neuronal apoptosis.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution and close monitoring for the efficacy of the anticholinesterase agent is recommended if it is to be used concomitantly with aminoglycosides, particularly in patients with myasthenia gravis. Concomitant use in these patients should be avoided if possible. If coadministration is considered necessary, the dose of the anticholinesterase agent may need to be increased. | ||||||||
