Details of Drug-Drug Interaction
| Drug General Information (ID: DDI1S3JQE2) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Isocarboxazid | Drug Info | Nesiritide | Drug Info | |||||
| Drug Type | Small molecule | Hormones | |||||||
| Therapeutic Class | Antidepressants | Recombinant Natriuretic Peptide | |||||||
| Structure | |||||||||
| Mechanism of Isocarboxazid-Nesiritide Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
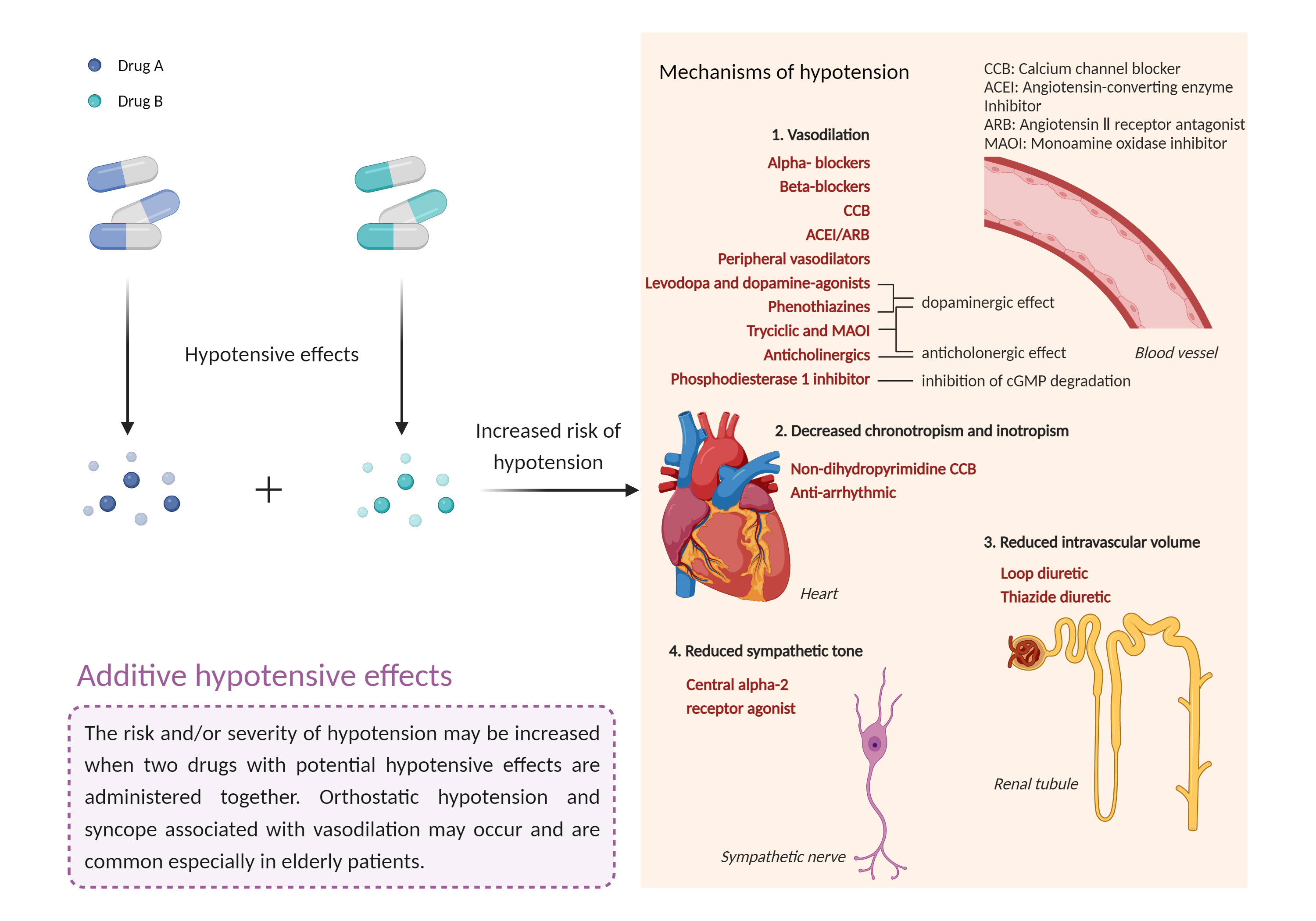
| Additive hypotensive effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Isocarboxazid | Nesiritide | |||||||
| Mechanism |
Hypotensive effects Monoamine oxidase non-selective Inhibitor |
Hypotensive effects Guanylate cyclase Agonist |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Monoamine oxidase | Structure Sequence | |||||||
| Protein Family | Flavin monoamine oxidase family | ||||||||
| Protein Function |
Catalyzes the oxidative deamination of primary and some secondary amine such as neurotransmitters, with concomitant reduction of oxygen to hydrogen peroxide and has important functions in the metabolism of neuroactive and vasoactive amines in the central nervous system and peripheral tissues (PubMed:20493079, PubMed:8316221, PubMed:18391214, PubMed:24169519). Preferentially oxidizes serotonin (PubMed:20493079, PubMed:24169519). Also catalyzes the oxidative deamination of kynuramine to 3-(2-aminophenyl)-3-oxopropanal that can spontaneously condense to 4-hydroxyquinoline (By similarity).
Click to Show/Hide
|
||||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Guanylate cyclase soluble | Structure Sequence | |||||||
| Protein Family | Adenylyl cyclase class-4/guanylyl cyclase family | ||||||||
| Protein Function |
There are two types of guanylate cyclases: soluble forms and membrane-associated receptor forms. Activated by nitric oxide in the presence of magnesium or manganese ions.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised during coadministration of MAOIs and other medications with hypotensive effects, especially during the first few weeks of treatment. Close monitoring for development of hypotension is recommended. Ambulatory patients should be advised to avoid rising abruptly from a sitting or recumbent position and to notify their physician if they experience dizziness, lightheadedness, syncope, orthostasis, or tachycardia. | ||||||||
