Details of Drug-Drug Interaction
| Drug General Information (ID: DDI1PSL7ZR) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Metformin | Drug Info | Capmatinib | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antidiabetic Agents | Multikinase Inhibitors | |||||||
| Structure | |||||||||
| Mechanism of Metformin-Capmatinib Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Transporter inhibition Click to Show/Hide Mechanism Graph | |||||||||
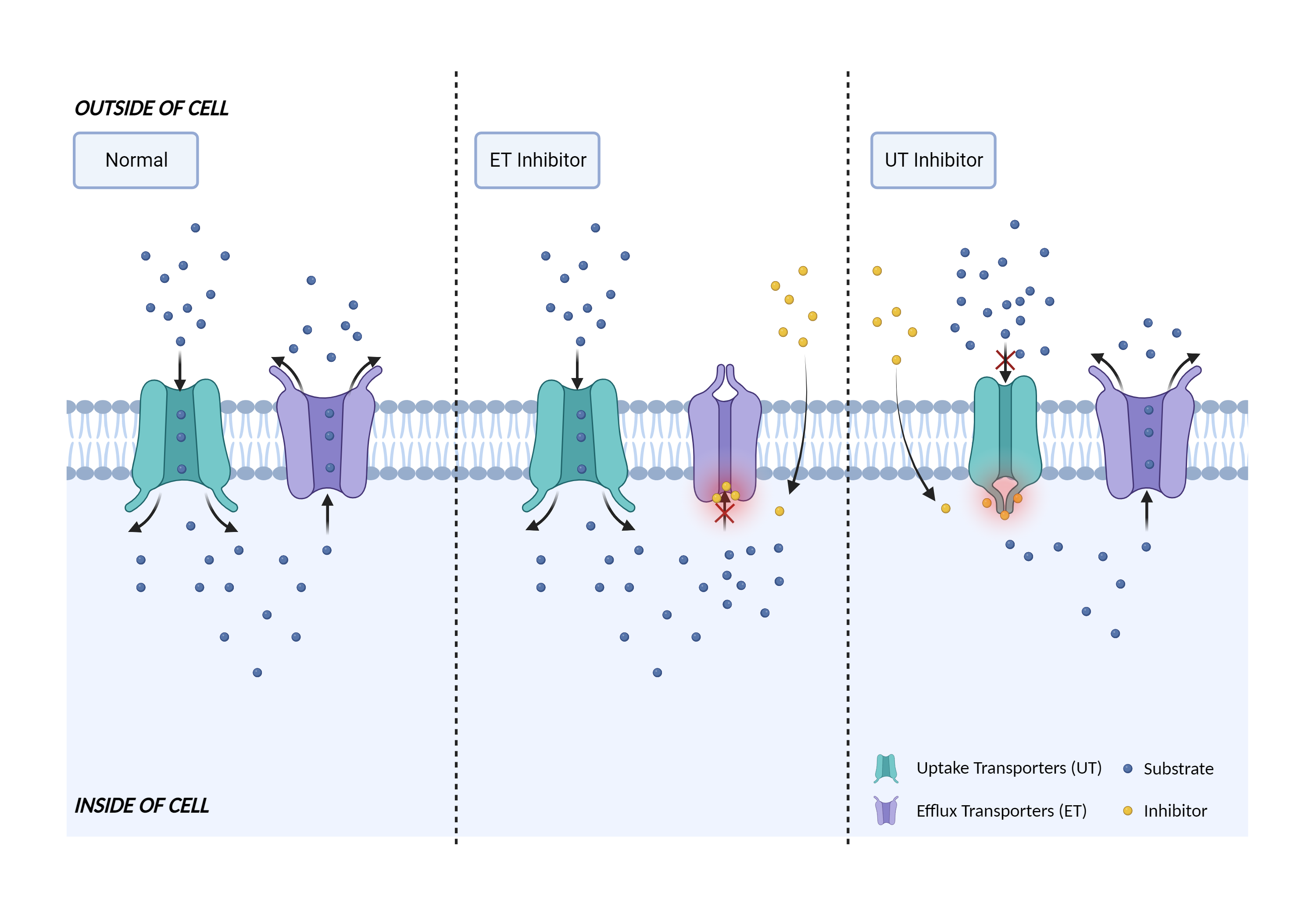 |
|||||||||
| Drug Name | Metformin | Capmatinib | |||||||
| Mechanism 1 | MATE1 substrate | MATE1 inhibitor | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Solute carrier family 47 member 1 |
×
Structure
Sequence
MEAPEEPAPVRGGPEATLEVRGSRCLRLSAFREELRALLVLAGPAFLVQLMVFLISFISSVFCGHLGKLELDAVTLAIAVINVTGVSVGFGLSSACDTLISQTYGSQNLKHVGVILQRSALVLLLCCFPCWALFLNTQHILLLFRQDPDVSRLTQTYVTIFIPALPATFLYMLQVKYLLNQGIVLPQIVTGVAANLVNALANYLFLHQLHLGVIGSALANLISQYTLALLLFLYILGKKLHQATWGGWSLECLQDWASFLRLAIPSMLMLCMEWWAYEVGSFLSGILGMVELGAQSIVYELAIIVYMVPAGFSVAASVRVGNALGAGDMEQARKSSTVSLLITVLFAVAFSVLLLSCKDHVGYIFTTDRDIINLVAQVVPIYAVSHLFEALACTSGGVLRGSGNQKVGAIVNTIGYYVVGLPIGIALMFATTLGVMGLWSGIIICTVFQAVCFLGFIIQLNWKKACQQAQVHANLKVNNVPRSGNSALPQDPLHPGCPENLEGILTNDVGKTGEPQSDQQMRQEEPLPEHPQDGAKLSRKQLVLRRGLLLLGVFLILLVGILVRFYVRIQ
|
|||||||
| Gene Name | MATE1 | ||||||||
| Uniprot ID | S47A1_HUMAN | ||||||||
| KEGG Pathway | hsa:55244 | ||||||||
| Protein Family | Multi antimicrobial extrusion (MATE) (TC 2.A.66.1) family | ||||||||
| Protein Function |
Solute transporter for tetraethylammonium (TEA), 1-methyl-4-phenylpyridinium (MPP), cimetidine, N-methylnicotinamide (NMN), metformin, creatinine, guanidine, procainamide, topotecan, estrone sulfate, acyclovir, ganciclovir and also the zwitterionic cephalosporin, cephalexin and cephradin. Seems to also play a role in the uptake of oxaliplatin (a new platinum anticancer agent). Able to transport paraquat (PQ or N,N-dimethyl-4-4'-bipiridinium); a widely used herbicid. Responsible for the secretion of cationic drugs across the brush border membranes.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Mechanism 2 | MATE2K substrate | MATE2K inhibitor | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Multidrug and toxin extrusion protein 2 |
×
Structure
Sequence
MDSLQDTVALDHGGCCPALSRLVPRGFGTEMWTLFALSGPLFLFQVLTFMIYIVSTVFCGHLGKVELASVTLAVAFVNVCGVSVGVGLSSACDTLMSQSFGSPNKKHVGVILQRGALVLLLCCLPCWALFLNTQHILLLFRQDPDVSRLTQDYVMIFIPGLPVIFLYNLLAKYLQNQGWLKGQEEESPFQTPGLSILHPSHSHLSRASFHLFQKITWPQVLSGVVGNCVNGVANYALVSVLNLGVRGSAYANIISQFAQTVFLLLYIVLKKLHLETWAGWSSQCLQDWGPFFSLAVPSMLMICVEWWAYEIGSFLMGLLSVVDLSAQAVIYEVATVTYMIPLGLSIGVCVRVGMALGAADTVQAKRSAVSGVLSIVGISLVLGTLISILKNQLGHIFTNDEDVIALVSQVLPVYSVFHVFEAICCVYGGVLRGTGKQAFGAAVNAITYYIIGLPLGILLTFVVRMRIMGLWLGMLACVFLATAAFVAYTARLDWKLAAEEAKKHSGRQQQQRAESTATRPGPEKAVLSSVATGSSPGITLTTYSRSECHVDFFRTPEEAHALSAPTSRLSVKQLVIRRGAALGAASATLMVGLTVRILATRH
|
|||||||
| Gene Name | MATE2 | ||||||||
| Uniprot ID | S47A2_HUMAN | ||||||||
| KEGG Pathway | hsa:146802 | ||||||||
| Protein Family | Multi antimicrobial extrusion (MATE) (TC 2.A.66.1) family | ||||||||
| Protein Function |
Solute transporter for tetraethylammonium (TEA), 1-methyl-4-phenylpyridinium (MPP), cimetidine, N-methylnicotinamide, metformin, creatinine, guanidine, procainamide, topotecan, estrone sulfate, acyclovir, and ganciclovir. Responsible for the secretion of cationic drugs across the brush border membranes.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Coadministration of capmatinib with drugs that are substrates of MATE1 and/or MATE2K should generally be avoided. However, if concomitant use is unavoidable, caution is advised, particularly with drugs that have a narrow therapeutic range. Clinical and laboratory monitoring should be considered whenever capmatinib is added to or withdrawn from therapy with these drugs, and the dosages adjusted as necessary. Patients should be monitored for the development of adverse effects. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Tabrecta (capmatinib). Novartis Pharmaceuticals, East Hanover, NJ. | ||||||||||||||||||
