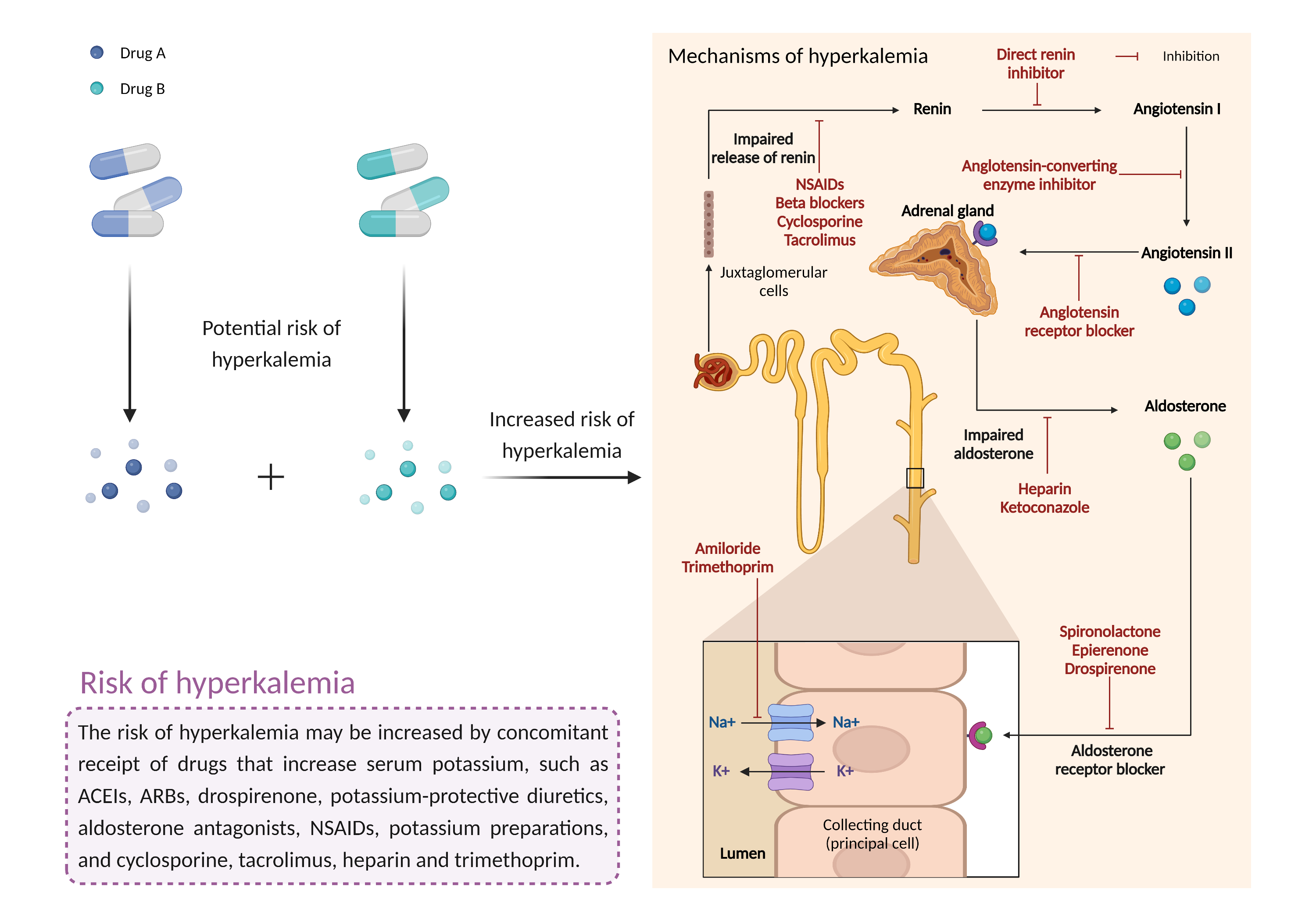
| References |
| 1 |
Product Information. Diovan (valsartan). Novartis Pharmaceuticals, East Hanover, NJ.
|
| 2 |
Product Information. Micardis (telmisartan). Boehringer-Ingelheim, Ridgefield, CT.
|
| 3 |
ONTARGET Investigators, Yusuf S, Teo KK, et al "Telmisartan, ramipril, or both in patients at high risk for vascular events." N Engl J Med 358 (2008): 1547-59. [PMID: 18700309]
|
| 4 |
Rossing K, Jacobsen P, Pietraszek L, Parving HH "Renoprotective effects of adding angiotensin II receptor blocker to maximal recommended doses of ACE inhibitor in diabetic nephropathy: a randomized double-blind crossover trial." Diabetes Care 26 (2003): 2268-74. [PMID: 12882847]
|
| 5 |
National Kidney Foundation "KDOQI Clinical Practice Guideline for Diabetes and CKD: 2012 update." Am J Kidney Dis 60 (2012): 850-86. [PMID: 23067652]
|
| 6 |
Jacobsen P, Andersen S, Rossing K, Jensen BR, Parving HH "Dual blockade of the renin-angiotensin system versus maximal recommended dose of ACE inhibition in diabetic nephropathy." Kidney Int 63 (2003): 1874-80. [PMID: 12675866]
|
| 7 |
MHRA. Medicines and Healthcare Regulatory Agency "Combination use of medicines from different classes of renin-angiotensin system blocking agents: risk of hyperkalaemia, hypotension, and impaired renal function--new warnings.".
|
| 8 |
Product Information. Atacand (candesartan). Astra Pharmaceuticals, Wayne, PA.
|
| 9 |
Cerner Multum, Inc. "UK Summary of Product Characteristics.".
|
| 10 |
EMA. European Medicines Agency "PRAC recommends against combined use of medicines affecting the renin-angiotensin (RAS) system: recommendation will now be considered by CHMP for final opinion." .
|
| 11 |
Laverman GD, Navis G, Henning RH, De Jong PE, De Zeeuw D "Dual renin-angiotensin system blockade at optimal doses for proteinuria." Kidney Int 62 (2002): 1020-5. [PMID: 12164886]
|
| 12 |
Pfeffer MA, McMurray JJ, Velazquez EJ, et al. "Valsartan, captopril, or both in myocardial infarction complicated by heart failure, left ventricular dysfunction, or both." N Engl J Med 349 (2003): 1893-1906. [PMID: 14610160]
|
| 13 |
McMurray JJ, Ostergren J, Swedberg K, et al. "Effects of candesartan in patients with chronic heart failure and reduced left-ventricular systolic function taking angiotensin-converting-enzyme inhibitors: the CHARM-Added trial." Lancet 362 (2003): 767-71. [PMID: 13678869]
|
| 14 |
Jacobsen P, Andersen S, Jensen BR, Parving HH "Additive effect of ACE inhibition and angiotensin II receptor blockade in type I diabetic patients with diabetic nephropathy." J Am Soc Nephrol 14 (2003): 992-9. [PMID: 12660333]
|
| 15 |
Guthrie RM "Review of ONTARGET: treating patients at high risk for vascular events with telmisartan, ramipril, or both. Commentary." Postgrad Med 121 (2009): 202-4. [PMID: 19332981]
|
| 16 |
Mann JF, Schmieder RE, McQueen M, et al. "Renal outcomes with telmisartan, ramipril, or both, in people at high vascular risk (the ONTARGET study): a multicentre, randomised, double-blind, controlled trial." Lancet 372 (2008): 547-53. [PMID: 18707986]
|