Details of Drug-Drug Interaction
| Drug General Information (ID: DDI1JUYW52) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Pegaspargase | Drug Info | Norgestimate | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antineoplastics | Contraceptive Agents | |||||||
| Structure | |||||||||
| Mechanism of Pegaspargase-Norgestimate Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
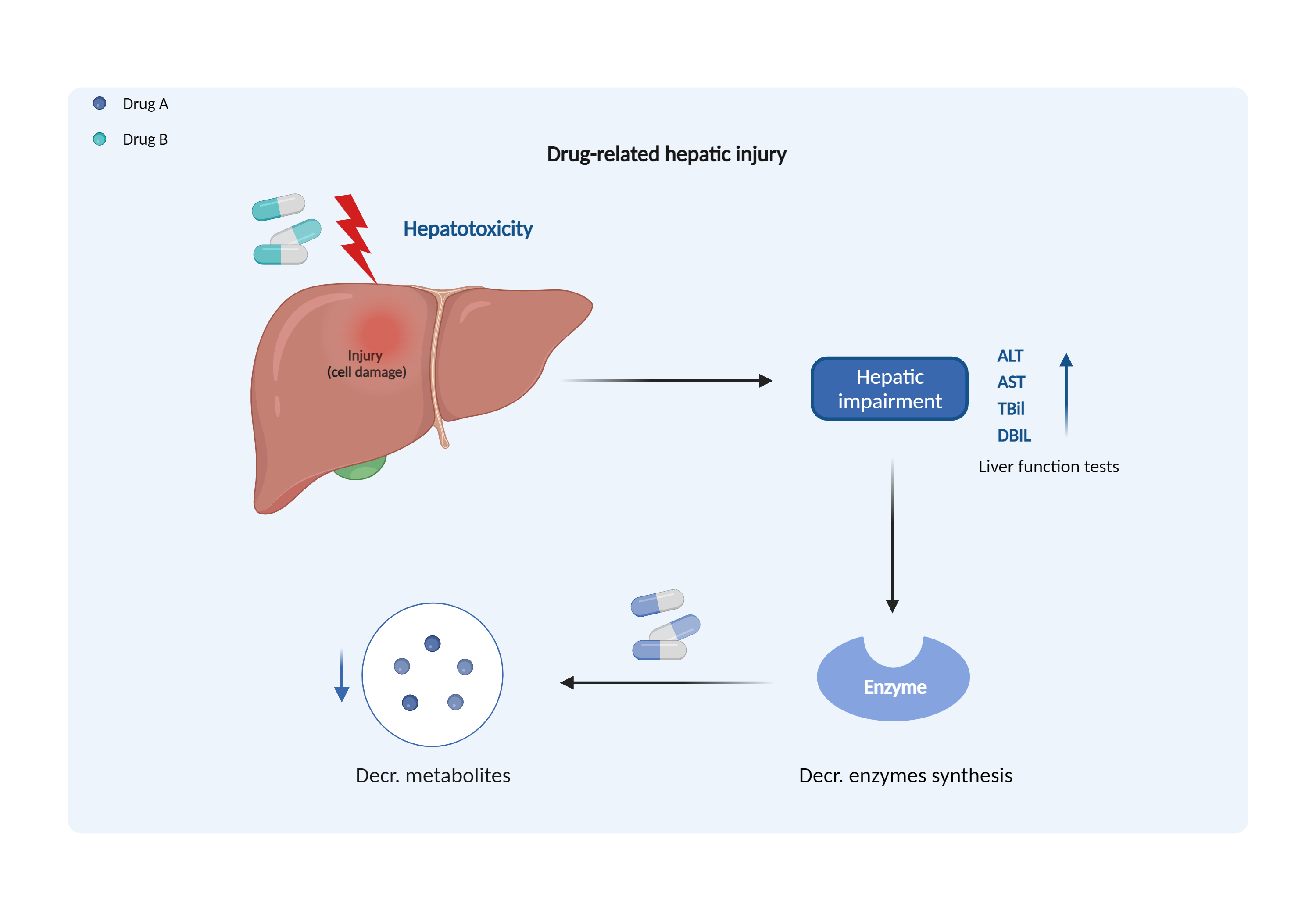
| Decreased metabolism due to hepatotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Pegaspargase | Norgestimate | |||||||
| Mechanism | Hepatotoxicity | Metabolized by the liver | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hepatotoxicity | ||||||||
| Factor Description | Drug-induced hepatotoxicity is an acute or chronic liver injury secondary to drugs. The metabolism of drugs is reduced when the liver is damaged and fewer enzymes are synthesized by the liver. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The use of oral contraceptives during asparaginase treatment is not recommended. Other non-oral contraceptive methods should be used in women of childbearing potential. According to the manufacturer, females of reproductive potential should be advised to use effective contraception during asparaginase treatment and for at least 3 to 6 months after the last dose. | ||||||||
