Details of Drug-Drug Interaction
| Drug General Information (ID: DDI1ED7QVX) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Oxcarbazepine | Drug Info | Dexlansoprazole | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Anticonvulsants | Proton Pump Inhibitors | |||||||
| Structure | |||||||||
| Mechanism of Oxcarbazepine-Dexlansoprazole Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| CYP450 enzyme inhibition Click to Show/Hide Mechanism Graph | |||||||||
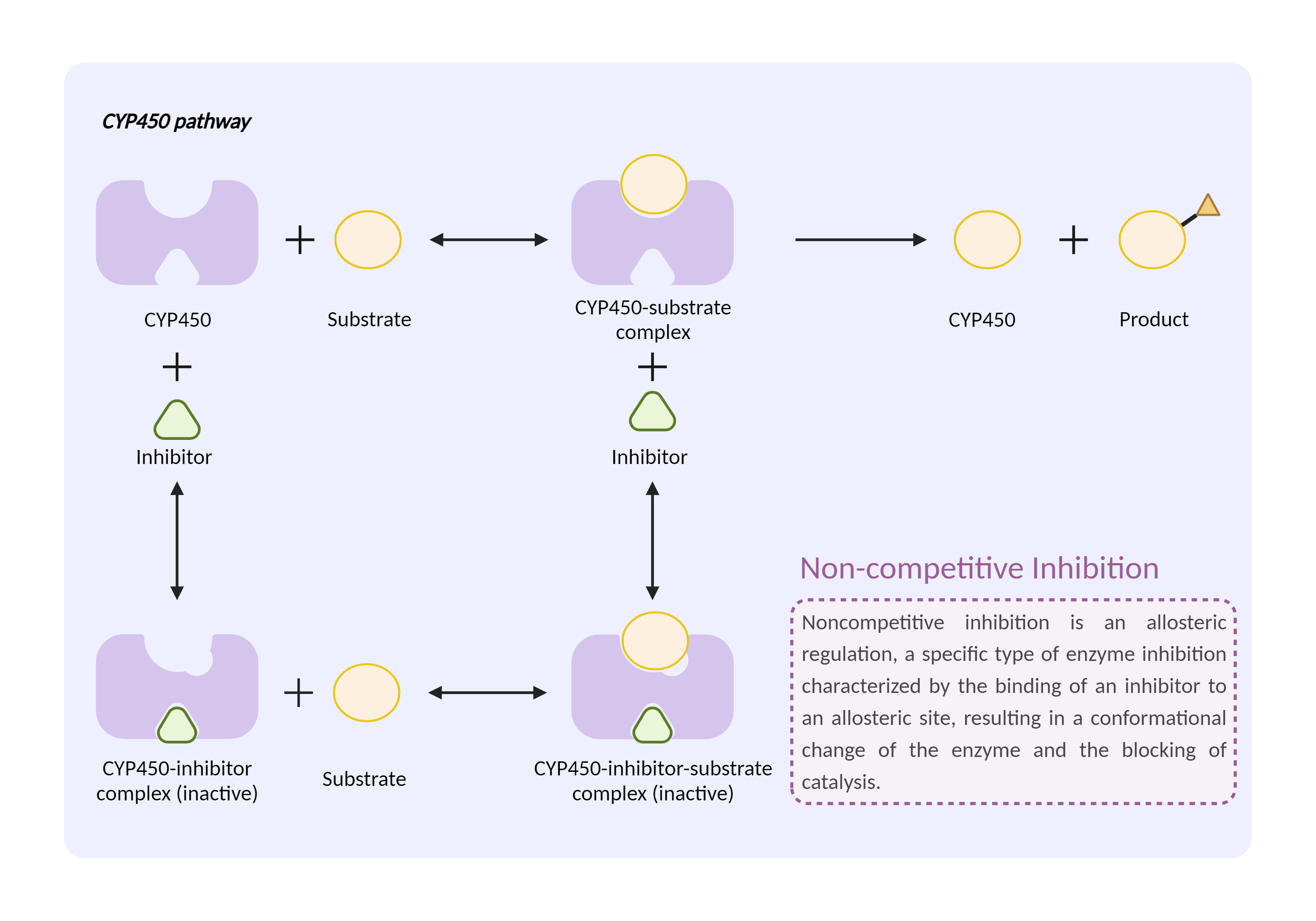 |
|||||||||
| Drug Name | Oxcarbazepine | Dexlansoprazole | |||||||
| Mechanism | CYP450 2C19 inhibitor | CYP450 2C19 substrate | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Cytochrome P450 2C19 |
×
Structure
Sequence
MDPFVVLVLCLSCLLLLSIWRQSSGRGKLPPGPTPLPVIGNILQIDIKDVSKSLTNLSKIYGPVFTLYFGLERMVVLHGYEVVKEALIDLGEEFSGRGHFPLAERANRGFGIVFSNGKRWKEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICSIIFQKRFDYKDQQFLNLMEKLNENIRIVSTPWIQICNNFPTIIDYFPGTHNKLLKNLAFMESDILEKVKEHQESMDINNPRDFIDCFLIKMEKEKQNQQSEFTIENLVITAADLLGAGTETTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRGHMPYTDAVVHEVQRYIDLIPTSLPHAVTCDVKFRNYLIPKGTTILTSLTSVLHDNKEFPNPEMFDPRHFLDEGGNFKKSNYFMPFSAGKRICVGEGLARMELFLFLTFILQNFNLKSLIDPKDLDTTPVVNGFASVPPFYQLCFIPV
|
|||||||
| Gene Name | CYP2C19 | ||||||||
| Uniprot ID | CP2CJ_HUMAN | ||||||||
| KEGG Pathway | hsa:1557 | ||||||||
| Protein Family | Cytochrome P450 family | ||||||||
| Protein Function |
A cytochrome P450 monooxygenase involved in the metabolism of polyunsaturated fatty acids (PUFA) (PubMed:18577768, PubMed:19965576, PubMed:20972997). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase) (PubMed:18577768, PubMed:19965576, PubMed:20972997). Catalyzes the hydroxylation of carbon-hydrogen bonds. Hydroxylates PUFA specifically at the omega-1 position (PubMed:18577768). Catalyzes the epoxidation of double bonds of PUFA (PubMed:20972997, PubMed:19965576). Also metabolizes plant monoterpenes such as limonene. Oxygenates (R)- and (S)-limonene to produce carveol and perillyl alcohol (PubMed:11950794). Responsible for the metabolism of a number of therapeutic agents such as the anticonvulsant drug S-mephenytoin, omeprazole, proguanil, certain barbiturates, diazepam, propranolol, citalopram and imipramine. Hydroxylates fenbendazole at the 4' position (PubMed:23959307).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if higher dosages of oxcarbazepine must be used concurrently with medications that undergo metabolism by CYP450 2C19, particularly those with a narrow therapeutic range. Dosage adjustments as well as clinical and laboratory monitoring may be appropriate for some drugs whenever oxcarbazepine is added to or withdrawn from therapy. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Cerner Multum, Inc. "Australian Product Information.". | ||||||||||||||||||
| 2 | Product Information. Trileptal (oxcarbazepine) Novartis Pharmaceuticals, East Hanover, NJ. | ||||||||||||||||||
