Details of Drug-Drug Interaction
| Drug General Information (ID: DDI08GPFZE) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Digitoxin | Drug Info | Milnacipran | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiarrhythmic Agents | Antidepressants | |||||||
| Structure | |||||||||
| Mechanism of Digitoxin-Milnacipran Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
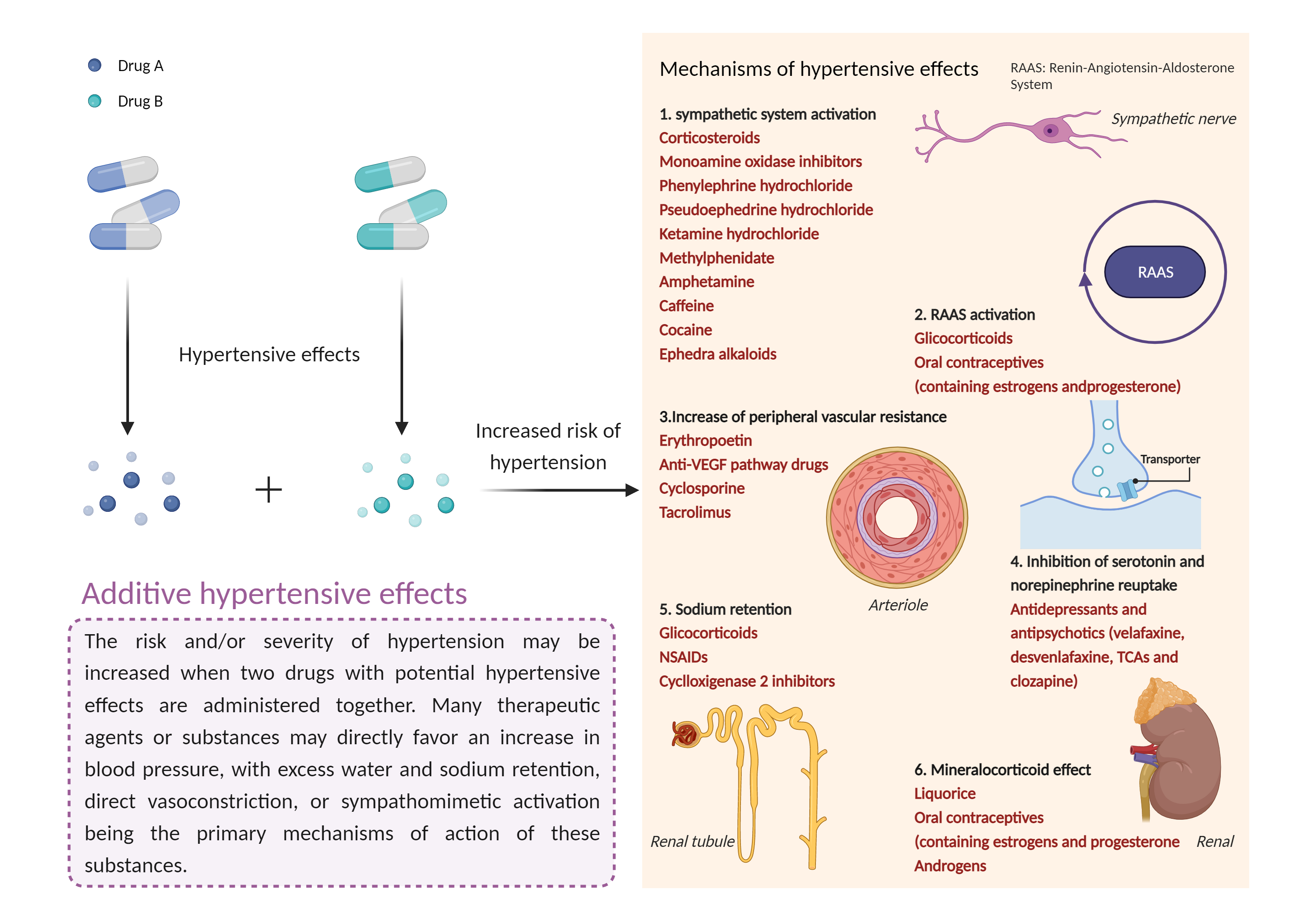
| Additive hypertensive effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Digitoxin | Milnacipran | |||||||
| Mechanism | Hypertensive effects | Hypertensive effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hypertensive effects | ||||||||
| Factor Description | Hypertension is defined as a blood pressure reading above a prescribed limit (130/90 mmHg). Most people with hypertension have no signs or symptoms; others may experience: severe headache, shortness of breath, nosebleeds, severe anxiety, and feeling a tingling sensation in the neck or head. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Coadministration of milnacipran and intravenous digoxin should be avoided. Caution is advised during coadministration with oral digoxin therapy. Blood pressure and heart rate should be measured prior to initiating milnacipran and periodically throughout treatment. A dosage reduction or discontinuation should be considered in patients who experience a sustained increase in blood pressure or heart rate while receiving milnacipran. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Savella (milnacipran). Forest Pharmaceuticals, St. Louis, MO. | ||||||||||||||||||
